Submission to the public consultation for the European Biotech Act
This response from the Centre for Future Generations (CFG) contributes to the European Commission’s public consultation for the European Biotech Act, launched on 4 August 2025. As a think-and-do tank supporting decision-makers in responsibly governing rapid technological change in the best interests of current and future generations, CFG welcomes the Commission’s ambition to strengthen Europe’s biotechnology ecosystem and innovation capabilities. Biotechnology is emerging as a strategic domain at the intersection of science, industry, and resilience – an engine for competitiveness and technological sovereignty. Its convergence with digitalisation, AI, and automation is giving rise to next-generation advanced biomanufacturing – where data and biology integrate to accelerate innovation and enable agile, intelligent production. Harnessing this transformation will require aligning Europe’s regulatory, infrastructural, and investment frameworks to turn scientific excellence into sustained industrial and societal advantage.
Comprehensive reform does not need to be achieved in a single step. What is essential is that the EU Biotech Act articulates a clear policy intent, enables transnational coordination, market appetite, regulatory commitment and defines priority areas for phased implementation. Such clarity would provide strategic direction to regulators, industry, and small innovators alike, ensuring that progressive adaptation can occur within a coherent and future-oriented framework.
Introduction: turning scientific excellence into strategic advantage
Biotechnology is a foundational, cross-cutting technology that drives innovation across health, industry, agriculture, and the green transition. Its systemic role makes it a strategic multiplier for industrial competitiveness, resilience, and technological sovereignty. Turning European biotechnology into strategic advantage requires a conscious shift from reactive and crisis-driven spending, towards sustained development of industrial capability: infrastructure, streamlined regulation, validated processes, and trained workforce that serve both continuous innovation and can pivot and scale during emergencies. This is achievable and economically sound.
Expanding biomanufacturing capacity and stockpiling alone will not secure strategic autonomy or guarantee the sustainable availability of medicines. True resilience depends on aligning capacity growth with reinforced innovation, regulatory coherence, and technology translation, creating an integrated ecosystem that connects discovery, scale-up, and industrial deployment. Rather than competing on raw volume, Europe can focus on high-value, knowledge-intensive biomanufacturing, building leadership in technologies that are harder to replicate, more agile, and innovation-driven. This would strengthen technological sovereignty in critical areas such as medical countermeasures (MCMs) and pharmaceuticals, while supporting biomanufacturing innovation and the broader bio-based economy.
Europe holds substantial biotechnological assets – 50 % of global exports in medical supplies and pharmaceuticals, supported by 25 % of global biologics capacity and over 4 million litres of installed bioreactor capacity[1]. Persistent translation gaps remain: SMEs often struggle to access pilot facilities, regulatory pathways remain fragmented, and strategic dependencies on imported active pharmaceutical ingredients continue to create vulnerabilities[2]. These dependencies are deepening and accelerating as biotechnology and AI converge. The newly announced Eli Lilly–NVIDIA “AI factory”[3] underscores the urgency of this transformation – integrating AI across discovery, design, and production – linking research, data infrastructure, and manufacturing intelligence into a single digital continuum. This transformation accelerates innovation but also amplifies reliance on external providers of foundational models, computational power, and secure digital systems, creating new layers of techno-industrial interdependence and biosecurity risk.
The scale-up valley of death
Biotechnology innovations reach a critical inflection point where scalability risk and costs surge as laboratory concepts move into pilot and demonstration phases.
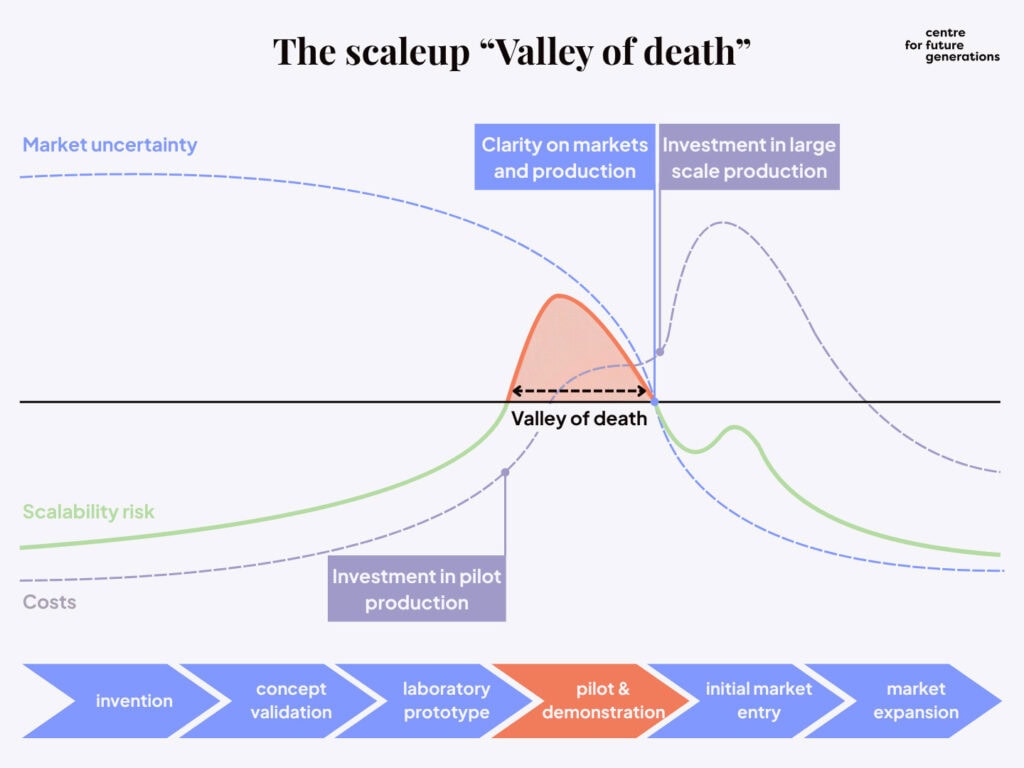
As shown in Figure 1, this stage – the ‘scale-up valley of death’ – is marked by steep rises in investment requirements and process uncertainty (scalability risk) before stabilising once manufacturing systems mature. Europe should act as an enabler at this point, expanding access to shared pilot and demonstration infrastructures and streamlining regulatory processes to de-risk scale-up.
Against this backdrop, this position paper examines how the EU Biotech Act can strengthen Europe’s strategic capabilities in advanced biomanufacturing. It focuses on the enabling regulatory (1. Biomanufacturing innovation & regulatory reform; 3. Regulatory Sandboxes) and scale-up (2. Technology Infrastructure) infrastructures that bridge innovation and industrial deployment, using medical countermeasures (MCMs) as a policy-relevant case study. The MCM lens illustrates how improving Europe’s biomanufacturing innovation ecosystem can enhance preparedness for new threats while addressing structural barriers to scaling and emphasizing the accelerating biosecurity concerns (4. Biosecurity) in a bio-digital biomanufacturing future.
Three priority actions for the EU Biotech Act
The EU Biotech Act is an opportunity to serve economic competitiveness, technological sovereignty and safeguard investment into the next generation of biotechnology infrastructures. It should:
- Establish a European biomanufacturing infrastructure network: federated pilot and demonstration facilities accessible to SMEs, bridging the scale-up ‘valley of death’
- Treat biomanufacturing capacity as strategic infrastructure: sustained public-private investment in facilities that serve commercial innovation during normal times and can surge to crisis response when needed
- Enable AI-integrated next-generation manufacturing: regulatory frameworks (platform authorisation, sandboxes, digital governance, AI lifecycle oversight) for intelligent, flexible and safe distributed production systems
Section 1. Disruptive advanced biomanufacturing innovation
Europe’s ability to translate modern biotechnology innovation into real-world impact depends on whether its regulatory and infrastructural systems evolve to support rapid advances in biomanufacturing. In 2022, Candelon et al. [5] forecast that synthetic biology would transform pharmaceuticals within five years and disrupt other biotechnology domains within ten. The rapid integration of mRNA manufacturing platforms, synthetic-biology-enabled systems accelerated by the COVID-19 response and later codified through European Medicines Agency and European Pharmacopoeia standards [6],[7], demonstrates how synthetic biology has and can fundamentally reshape pharmaceutical manufacturing – shifting it towards platforms, modularity, flexibility, and rapid scalability[8]. Before 2019, mRNA research produced only about 571 scientific papers; by 2024, that figure had increased more than fourteen-fold, with over 280 vaccine candidates in development – nearly half already in clinical stages and most targeting diseases beyond COVID-19 (Fig.2)[9].
mRNA platforms – disruptive biotechnology in practice
However, disruption has been uneven across biotechnology sectors and modalities. Differences in technological maturity, investment concentration, and fragmented governance have created divergent trajectories[10],7. Innovative synthetic-biology biomanufacturing often spans pharmaceutical, environmental and GMO frameworks, navigating overlapping regimes, fragmented implementation and duplicated assessments. Currently there is no clear regulatory path to recognise a biomanufacturing platform itself—the underlying, adaptable process—because approval remains tied to an authorised product. Emerging precedents, such as the European Medicines Agency draft guideline for mRNA vaccines, reference “platform technology/prior knowledge”. But no coherent cross-sector framework exists at the European level [11],[12],[13]. large firms this uncertainty introduces late-stage regulatory risk, deterring strategic investment in disruptive manufacturing systems. For SMEs it means high compliance costs and scaling barriers, as regulatory routes remain opaque and unpredictable. Meanwhile, investors redirect capital to technologies with clearer pathways to market and lower regulatory risk.
During the COVID-19 pandemic the EU showed remarkable regulatory agility through emergency-use authorisations and adaptive oversight, laying the groundwork for later codification of these mRNA platform standards in 2023–2025. This shows that Europe can act swiftly in crisis yet lacks anticipatory, future-proof frameworks. Equally decisive were the industrial and infrastructural responses: manufacturing scale-up drew on repurposed GMP facilities and modular build-up of flexible production capacity, exemplified by Pfizer-BioNTech’s distributed manufacturing network[14]. Firms accepted unprecedented technical and financial risk to innovate in real time, underpinned by large-scale public procurement that de-risked investment in novel biomanufacturing platforms[15]. Such exceptional conditions cannot serve as Europe’s default innovation model, the EU will need to strengthen its role as a market shaper beyond an investor. The strategic challenge is to replicate that alignment in normal times – through bold anticipatory regulation, open-access pilot infrastructure, proactive market shaping, and predictable funding that make innovation translation routine..
Traditional biologics manufacturing develops a bespoke process for each product, with regulations requiring separate assessment and authorisation. Platform-based biomanufacturing reverses this logic. A single validated system (mRNA, viral-vector, or cell-free expression) generates multiple products by altering programmable inputs whilst maintaining constant core processes. These platforms rely on digital process control, iterative optimisation, and standardised quality frameworks.
Europe’s regulatory and infrastructure systems remain product-specific, limiting platform scalability. A paradigm shift is needed: from regulating products individually to enabling platforms collectively, and from static capacity to pre-qualified, flexible infrastructure capable of rapid mobilisation.
The Biotech Act should establish frameworks that pre-qualify modular platforms before emergencies, embed them in joint European infrastructure (section 2: Technology Infrastructures), and enable rapid deployment for both crisis response and routine production.
Advanced biomanufacturing for economic and health resilience
Ongoing breakthroughs must be translated into new medical countermeasures (MCMs) fast enough to counter today’s threats. This chapter uses MCMs to demonstrate why platform-based regulation is essential for European biotechnology. Four case studies: AI-orchestrated cell-free synthesis, plant molecular farming, bacteriophage manufacturing, and non-traditional host platforms illustrate how current product-centric regulation prevents promising biomanufacturing innovations from reaching scale. The EU Biotech Act can enable this paradigm shift towards streamlined, platform-based frameworks.
Case study 1
AI-orchestrated cell-free protein synthesis; 8-hour production, 2-year regulatory timelines
Cell-free protein synthesis (CFPS) uses cellular extracts, rather than living cells, to produce proteins directly from DNA or RNA templates. Production occurs within 8-10 hours rather than weeks[16]. Extracts can be freeze-dried for ambient storage and shipping, enabling modular, distributed biomanufacturing. CFPS has produced antibody fragments, vaccines, anti-cancer agents, human interferon alpha-2b, enzymes, viruses, and virus-like particles (VLPs), with several candidates advancing through clinical trials[17].
AI-orchestrated CFPS (AI-CF) integrates machine learning models trained on process data to optimise reaction parameters (enzyme ratios, codon usage, buffer conditions), maximising reproducibility, yield, and solubility. AI “bio-compilers” convert digital DNA or RNA sequences into standardised manufacturing recipes, supporting 3-7-day sequence-to-protein timelines and simultaneous multi-site production[18]. The underlying biology remains the same, but the regulatory focus shifts from traditional site-based process validation to digital governance, AI model lifecycle oversight, and AI quality control authorisation[19],[20],[21].
CFPS as biomanufacturing innovation for distributed resilience
Short DNA-to-protein timelines enable rapid, localised therapy and diagnostics production during outbreaks. CFPS can produce complex or cytotoxic proteins that are challenging to make in living cells. Its direct scalability supports both microscale prototyping, and recently, up to 4,500 L GMP production – both R&D and emergency-scale deployment[22]. Freeze-dried extracts reduce cold-chain dependencies and enable stockpiling across Member States[23]. AI-CF enhances lot-to-lot reproducibility and automates regulatory documentation (digital batch records, recipe passports). Freeze-dried kits deployed at pre-clinical or clinical sites could support adaptive, decentralised trials during outbreaks when manufacturing and logistics are disrupted.
CFPS and AI-CF sit at the intersection of medicinal product law, GMO contained use regulations, the EU AI Act, and GMP Annex 11, yet no integrated pathway exists to authorise them as manufacturing platforms. Clear platform-level authorisation processes (covering freeze-dried CFPS kits, digital compilers, AI lifecycle master files, and digital recipe passports) would streamline approvals and enable distributed deployment. This reduces centralised capital expenditure whilst creating flexible surge capacity at regional or hospital level, strategically important for MCMs. By regulating CFPS and AI-CF as evolving platforms rather than individual products, advancing a key new mechanism in adaptive governance.
Case study 2
Plant molecular farming; rapid scale-out in closed greenhouses
Plant molecular farming (PMF) uses plants, commonly Nicotiana benthamiana, as contained bioreactors to produce complex biologics (VLP vaccines, monoclonal antibodies) by inserting DNA sequences that instruct plants to synthesise target proteins for a limited period. Initial material can be obtained within 2-6 weeks after sequence availability, using greenhouse or plant-factory facilities rather than stainless-steel bioreactors[24],[25],[26].
Rapid scaling for MCMs
Modular greenhouses enable rapid scale-out across Member States for regional surge capacity. Production in non-human hosts reduces contamination risks and supports diverse product types. A plant-derived VLP COVID-19 vaccine achieved 71% efficacy in clinical trials and regulatory approval in Canada[27]. However, Medicago’s vaccine reached the Canadian market only after mRNA platforms had saturated demand, illustrating how regulatory and manufacturing timelines determine commercial viability[28]. In the EU, current GMO contained-use and environmental authorisation timelines (12–24 months in some Member States) create regulatory barriers for PMF platforms.
PMF requires lower capital investment than mammalian cell facilities and can leverage Europe’s agricultural infrastructure for distributed, flexible capacity [29],[30]. Harmonised contained-use procedures, platform-level dossiers (covering host, vector, and purification strategies), and integration into HERA/EU-Fab frameworks would enable PMF to support emergency response and SME-led innovation.
AI-automated plant factories: a biomanufacturing future
Fully enclosed greenhouses where AI-controlled robots manage light, humidity, and nutrients with molecular precision represent the convergence of agriculture, robotics, and biotechnology. AI-driven sensors monitor growth in real time, adjusting conditions to maximise yield and quality whilst learning from every production cycle. In emergencies, digital “recipes” could be transmitted across Member States, enabling identical batches to be grown simultaneously within days – a living, distributed manufacturing network.
This vision shows how the EU’s agricultural and digital assets could converge into a new class of bio-robotic production facilities[31],[32],[33]. Realising this vision requires updated GMP and AI oversight frameworks, but offers a glimpse of Europe’s possible next-generation biomanufacturing frontier.
Reflection: Positioning platforms for sustainable use
Several of the technologies described – such as cell-free protein synthesis and plant molecular farming – offer clear advantages for rapid response, yet their long-term sustainability depends on how they are positioned and utilised within the broader market landscape.
It will be important to examine how these platforms can be aligned with ongoing manufacturing needs and market demand, ensuring that their use extends beyond emergency deployment. The experience of Samabriva, a French biotechnology company using plant root-based bioproduction systems for active pharmaceutical ingredients (APIs), demonstrates how positioning plant-derived technologies within established value chains can create continuous, commercially viable utilisation while maintaining responsiveness for surge production.
Further study could clarify how appropriate business models, partnerships, and policy frameworks might support the continued use and readiness of such platforms. Sustained application would help maintain availability, skilled capacity, and operational continuity for rapid mobilisation when required.
Case study 3
AI-enabled bacteriophage therapy, adaptive manufacturing, fragmented regulation
Bacteriophages are viruses that infect and destroy specific bacteria. Therapeutic preparations are produced as standardised cocktails or patient-specific magistral medicines in hospital pharmacies within 5–14 days of pathogen identification. The 2024 European Pharmacopoeia introduced harmonised standards for phage products, yet national implementation remains inconsistent. Conventional GMP frameworks struggle to accommodate iterative, hospital-based production. Bacteriophage therapy illustrates a new pathway in biomanufacturing innovation – adaptive, and decentralised rather than industrial in scale.
Phage manufacturing depends on precise control of process parameters and real-time monitoring of purity and potency[34]. AI-driven analytics can predict deviations, optimise host–phage ratios, and automate documentation, transforming phage preparation into data-driven, decentralised biomanufacturings[35],[36]. Bacteriophage therapy is not a mass-production breakthrough but a regulatory and digital innovation that exposes Europe’s fragmented governance of adaptive biomanufacturing – and shows how AI-enabled quality assurance could close that gap.
Phages as MCMs
Phages can be rapidly matched to resistant infections, supporting EU antimicrobial resistance priorities. Hospital-based production enables near-patient manufacture during outbreaks, reducing supply-chain dependence. Importantly, phage platforms could also be considered as adaptable MCMs for non-traditional and emerging biological threats, including engineered pathogens or “mirror-life” constructs developed through xenobiology or synthetic-biology pathways. Phage modularity, specificity, and scalability position them as a potential “plug-in” platform for rapid response and targeted containment.
Beyond xenobiological scenarios, phages align with broader biodefence and health-security MCM frameworks, offering flexible antibacterial and biocontrol capacities relevant to Chemical, Biological, Radiological and Nuclear (CBRN) preparedness and pandemic-scale resilience. Given their capacity for benign and hostile application alike, phages and phage-derived tools should also be recognised as dual-use technologies, requiring governance models that balance innovation, rapid deployment, and biosecurity safeguards.
Belgium has implemented a formal magistral pathway; elsewhere, divergent interpretations of Directive 2001/83/EC and overlapping GMO and GMP rules hinder uptake. Phage therapy requires platform-level governance covering shared phage libraries, digital quality assurance systems, and data standards. Integrating AI-based process control into GMP Annex 11 and AI Act frameworks could modernise oversight for adaptive, small-batch biologics and transform regulatory innovation into an additional tool in the box for responding to chemical, biological, radiological, or nuclear (CBRN) threats, or emerging infectious diseases.
Phages beyond MCMs: harnessing precision biocontrol across sectors
Phages are being developed as precision biocontrol agents in crops, livestock, and food processing, and for managing microbial populations in disease-carrying insects[37],[38]. They offer antibiotic-free, residue-free alternatives aligned with EU One Health and sustainable biotechnology priorities.
Engineered phages are also being explored as biological vectors for drug and gene delivery, targeting specific cells or microbiome niches[39],[40],[41]. These applications illustrate the market potential for commercial phage platforms as modular biological chassis bridging therapeutic, industrial, and environmental domains.
Whilst large-scale bioreactor production of phages is technically achievable, deployment remains limited by variability in phage–host systems and the absence of harmonised EU quality standards. Fragmented regulation across medicinal, veterinary, and biocontrol frameworks continues to deter coordinated industrial participation and long-term capital investment.
Regulatory actions to enable platform-based biomanufacturing
The EU Biotech Act provides an opportunity to modernise regulations designed for single-site, static, cell-based processes. Four emerging platform categories set out in this section, and non-traditional platforms (insect and plant cell systems) – are maturing technically but face regulatory frameworks that generate delays, duplication, and uncertainty. Targeted reforms can align EU regulation with these technologies whilst maintaining high standards.
1. Platform technology master files (PTMFs)
Establish Platform Technology Master Files (PTMFs) to enable regulators to assess the biomanufacturing platform – covering design, validation, and quality systems – once. Subsequent products would reference this authorisation, focusing only on product-specific attributes. PTMFs should be managed through a lifecycle evaluation approach, allowing regulators to incorporate real-world operational evidence and continuous platform improvements. This mirrors the post-COVID mRNA regulatory model, reducing timelines without lowering standards.
2. Redefine “batch” for distributed manufacturing
Update regulatory definitions of “batch” to reflect distributed and continuous manufacturing models. In systems such as AI-CFPS, PMF, and PHG, production relies on centrally certified upstream materials (e.g. freeze-dried extracts, plant inocula, phage banks) distributed across multiple sites. Under defined quality and traceability conditions, the regulatory batch should correspond to certified upstream starting material rather than each local run, supported by data-driven traceability tools that provide continuous oversight across sites.
3. Apply risk-based and adaptive testing strategies
Testing requirements should reflect the realities of decentralised and evolving biomanufacturing systems. Regulatory evaluation should distinguish between:
- Platform-level testing of stable elements (extract quality, compiler performance, plant containment, phage library characterisation); and
- Product-specific testing of clinical attributes (potency, purity, identity).
A risk-based and adaptive testing framework would focus analytical effort where variability is highest, supported by continuous performance monitoring and real-world evidence (RWE) to inform lifecycle regulatory decisions.
4. Harmonise contained-use and GMO interpretation
Member States interpret contained-use and GMO regulations inconsistently, especially for low-risk or inactivated materials. Establishing proportionate, harmonised, and mutually recognised procedures, grounded in a common EU risk-based framework, would remove duplication, accelerate technology transfer, and support continuous, cross-border operations.
A harmonised approach should include shared data systems to enable transparent, ongoing regulatory visibility across Member States.
5. Enable adaptive authorisation with real-world evidence
Rapidly evolving biomanufacturing platforms require adaptive regulatory models that evolve with technology. The Biotech Act should promote adaptive qualification pathways combining initial platform authorisation with continuous assessment informed by real-world operational data—including evidence from emergency use, compassionate access, and distributed manufacturing performance. This lifecycle model would enable regulators to maintain high safety standards while accommodating iterative technological improvements.
6. Cyber-biosecurity and biosecurity-by-design
Rapidly digitising biomanufacturing platforms depend on interconnected software, data pipelines, and physical processes. A proportionate cyber-biosecurity approach (combining classical biosecurity with cybersecurity controls for digital–biological interfaces) should be embedded at platform level rather than added product by product.
Technical measures for securing AI-integrated biomanufacturing could include, from[42]:
- Establish trusted reference datasets with traceable provenance for AI training and validation
- Implement hardware verification programmes (analogous to Federal Communications Commission Internet of Things device certification)
- Embed redundancy (duplicate critical systems and distributed capacity across sites) as primary strategy to prevent single-point failures from halting production
- Adopt AI safety testing methodologies to validate safe operational bounds for autonomous systems
- Develop new threat models for software supply chains in bioeconomy beyond traditional cybersecurity practices
7. Integrate AI model lifecycle management
AI-enabled compilers and process control systems evolve through retraining and optimisation. AI Model Lifecycle Master Files should document training data, validation methods, and update protocols once, allowing manufacturers to reference them across submissions. This would give regulators real-time visibility into evolving AI models while enabling adaptive oversight of algorithmic performance, aligned with the EU AI Act and GMP Annex 11.
8. Establish a digital recipe passport system
Create an EU-level Digital Recipe Passport linking each product sequence or variant to its compiler version, site, and batch data. This system should function as a continuous data layer for regulatory oversight, enabling rapid registration, traceability, pharmacovigilance, and targeted recalls. It would allow real-time regulatory access to key process data, aligning authorisation with the lifecycle and evidence-driven framework of the Biotech Act.
9. Update guidance for emerging platforms
Outdated regulatory guidance creates uncertainty and slows technology uptake. The Commission, in coordination with EU agencies, should issue updated, adaptive guidance clarifying classification, comparability, and lifecycle evidence expectations for novel systems (e.g. plant cell suspensions, algae, engineered phages). Such non-legislative instruments should explicitly recognise continuous data use, digital process monitoring, and adaptive qualification as part of modern regulatory practice.
10. Integrate platform regulation into EU preparedness frameworks
Pre-certified, continuously qualified platforms should be embedded in HERA’s preparedness and emergency frameworks, ensuring rapid deployment of validated technologies during crises. Integration should include digital traceability systems, real-world performance data flows, and adaptive review mechanisms to enable coordinated oversight and flexible scaling across Member States. This would link Europe’s biomanufacturing preparedness directly to its adaptive and evidence-driven regulatory ecosystem.
Conclusions
Common regulatory barriers, coordinated solutions
Innovative biomanufacturing platforms share common regulatory barriers: fragmented platform recognition, misaligned batch definitions, disproportionate testing requirements, divergent GMO interpretations, and absent adaptive authorisation pathways. They also share a critical vulnerability: dependence on interconnected digital–biological interfaces that current frameworks do not adequately secure.
Securing the cyber-bio interface must be a foundational priority. As biomanufacturing becomes increasingly digital, automated, and distributed, cybersecurity is integrated at the design stage of platform development. Without embedded cyber-biosecurity, investments in advanced platforms risk creating sophisticated but exploitable systems.
Targeted, proportionate, and harmonised reforms could enable the EU to unlock the full potential of innovative biomanufacturing platforms while maintaining high regulatory standards and positioning Europe as a global leader in next-generation biomanufacturing.
Section 2. Technology infrastructures for scaling innovation to market
From research excellence to biomanufacturing leadership
The EU faces a persistent “scale-up valley of death” (Fig.2[43]) between prototype and commercial manufacturing. CFG’s industry engagement exercise identified limited pilot capacity, regulatory misalignment, and uneven SME access as principal barriers to integrated biotechnology ecosystems[44]. Some SMEs face 18-24 month delays for pilot-GMP access, narrowing patent windows and undermining competitiveness.
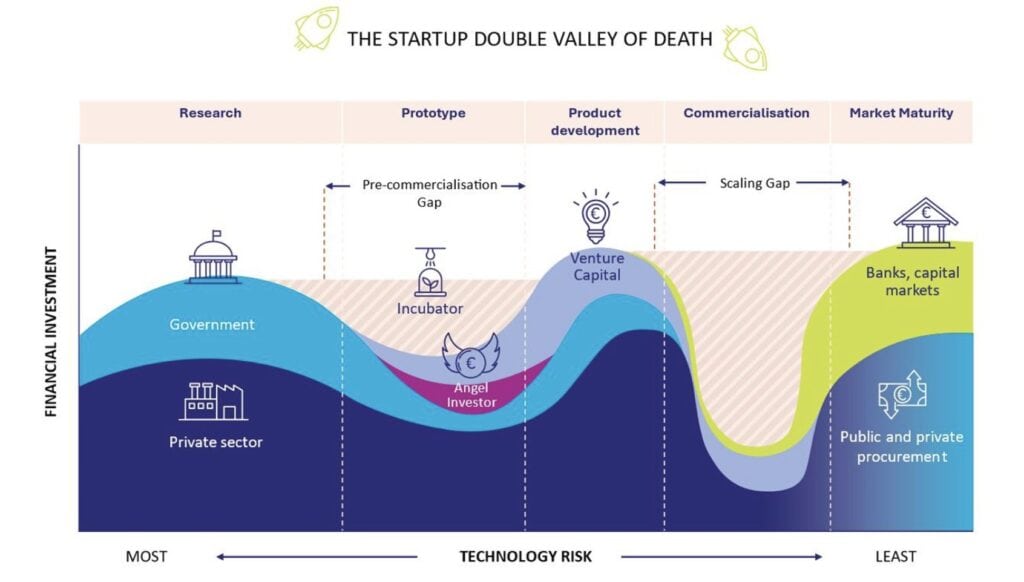
Embedding advanced biomanufacturing pilot infrastructures within the EU Biotech Act, analogous to pilot lines in semiconductors and photonics[45]), would directly address this structural deficit[46],[47],[48] – connecting R&D excellence to industrialisation whilst building sovereign capability in next-generation biotechnology.
Large segments of cost-sensitive production will continue to be more economically located outside the EU, reflecting legitimate cost-base advantages. The strategic concern lies elsewhere: Europe risks losing access to its own EU-developed, high-value biotechnologies because it lacks the pilot-scale infrastructure, regulatory flexibility, and investment coordination required to industrialise them domestically. Our CFG industry listening exercise[49] revealed that some SMEs face 18-24 month delays for pilot-GMP access, narrowing patent windows and undermining competitiveness [50].
Global biomanufacturing is shifting externally: in-house production declined from 57.6% (2006) to 29.7% (2023), with 67.8% of firms planning offshore operations within five years6. As biomanufacturing shifts away from in-house models toward external, pilot-based, and contract structures, Europe must ensure that its innovators can scale domestically rather than seeking capacity abroad or never commercialising. High-value outsourcing concentrates in the United States (47.8%) and Germany 6. This underscores Europe’s need to sustain its position by strengthening strategic innovation-critical stages through the expansion of pilot-scale and demonstration infrastructures.
Other jurisdictions act decisively: UK invested £151 million in viral-vector and DNA pilot facilities[51]; Singapore integrates pilot-to-commercial capacity through Biopolis[52] and the US PCAST Report (2022) highlights national pilot-capacity shortfalls calling for new biomanufacturing hubs – a gap Europe could leverage to attract investment and strengthen its leadership[53].
The subsequent 2025 U.S. Biotech Strategy report to Congress[54] identifies limited pilot and production capability, particularly in biopharmaceuticals and advanced therapeutics, as a national vulnerability and recommends coordinated federal action to expand scale-up capacity. It calls for the Departments of Energy and Commerce to establish a national network of pre-commercial manufacturing facilities to bridge the scale-up gap and strengthen domestic production. The report further advises that biotechnology infrastructure and data be designated as part of U.S. critical infrastructure, reflecting their strategic value for economic resilience and security.
The logic underpinning dedicated biomanufacturing hubs in biotechnology applies equally to other emerging deep-tech sectors. Neurotechnology, in particular, embodies strong societal relevance and industrial promise, yet progress is hindered by the high cost and technical complexity of specialised R&D infrastructure that most SMEs cannot access. As underlined in Wired for the Future – Boosting Europe’s Neurotechnology Frontier (CFG, 2024)[13] and illustrated by initiatives such as the Open Innovation Test Beds[14] , the creation of a shared, pre-competitive European platform providing validated and modular components could effectively bridge this gap. Focused investment in such cross-cutting infrastructure would facilitate access to innovation resources, accelerate clinical and industrial translation, and consolidate Europe’s leadership in advanced manufacturing and next-generation health technologies.
Technology infrastructures as bridges from R&D to industrial deployment
Europe’s biotechnology ecosystem comprises Research Infrastructures (RIs), which excel in discovery but not process scale-up, and Contract Development and Manufacturing Organisations (CDMOs), which scale established technologies whilst avoiding unproven approaches. This leaves emerging biotechnologies without translation environments.
Technology infrastructures (TIs) fill this gap by providing shared, pre-commercial environments for process innovation and regulatory maturation. TIs include pilot lines, pilot plants, demonstration facilities, test-beds, and living labs. Industry uses them for developing new technologies (77%), testing in real-world conditions (65%), manufacturing-scale trials (60%), and workforce development (61%)[56]. Their role is complementary to CDMOs: they operate upstream, where technological and regulatory uncertainty is greatest, to enable faster, lower-risk industrial scaling.
However, 70% of SMEs report financial constraints as the largest barrier to TI access, compared with under 50% of large enterprises[57]. Industry currently engages with TIs primarily through university-based infrastructures, indicating academic rather than industrial orientation. To address this, dedicated SME voucher schemes, industry-facing access pathways, and public–private partnership models should complement the development of new hubs and specialised sites. This would ensure that Europe’s TIs are both accessible and commercially responsive, enabling SMEs to translate innovation into scalable biomanufacturing more effectively.
The Biotech Act as the sector-specific implementation of TI strategy
EMA Executive Director Emer Cooke has said that addressing medicines shortages and dependency risks calls for an “infrastructure policy solution” rather than adjustments limited to the pharmaceutical legislative framework[58]. The European Strategy on Research and Technology Infrastructures (September 2025) provides a strong framework, but broad horizontal strategies cannot deliver tailored governance and investment mechanisms. The EU Biotech Act offers the vehicle to apply this framework concretely within biotechnology, anchoring pilot infrastructures as strategic assets. Current TI funding remains fragmented between EU and national levels, with most infrastructures relying on short-term competitive calls[59],[60]. A sustainable public–private partnership model combining EU coordination, national co-funding, and private participation would work as market enablers to enhance viability and industrial engagement.
A coherent TI implementation strategy should be:
Sector-specific, aligned with Europe’s strategic priorities and sector-specific needs;
- Legislatively codified, to anchor governance, funding, and regulatory alignment;
- Financially inclusive, for example through SME vouchers addressing primary access barriers;
- Digitally integrated, via a single-entry interface for industry access.
- Dedicated regulatory environments enabling controlled experimentation
The European Strategy Forum for Research Infrastructures (ESFRI) framework offers a successful governance model for research infrastructures that could be adapted for TIs – with biotechnology and biomanufacturing as the pilot domain. These principles form the basis of our core infrastructure pillars.
Core infrastructure pillars for EU technology infrastructures
A coherent pan-European biomanufacturing network requires five interconnected pillars:
Biofoundries integrate robotics, AI-driven design, and advanced analytics to operationalise the Design–Build–Test–Learn cycle. They enable iterative refinement of strains, pathways, and production parameters under industrially-relevant conditions, bridging conceptual innovation and reproducible process performance.
Modular pilot and demonstration facilities provide intermediate-scale environments (100–5,000 L) for process validation, regulatory-grade data generation, and early clinical manufacturing. Co-location with clinical and regulatory ecosystems anchors early-stage trials in Europe and reduces SME capital barriers.
Regulatory sandboxes are structured environments where innovators, regulators, and infrastructure operators test new manufacturing technologies and regulatory approaches in parallel. They enable early identification of regulatory requirements, data generation for guidance, and platform qualification. Embedding sandboxes within TIs ensures regulatory learning occurs in operational contexts not solely through theoretical consultation or post-approval adaptation.
Integrated digital data environments unify process and analytical data, delivering real-time traceability through digital recipe passports linking sequences, batches, and sites. Aggregating anonymised datasets creates collective process intelligence for machine learning-driven optimisation and facilitates regulatory integration through continuous verification. This builds federated biomanufacturing intelligence – a shared digital backbone providing competitive advantage.
Nvidia Flare is one of the newly emerging open-source frameworks for federated learning, designed to enable secure, multi-partner collaboration without sharing raw data[61]. Although originally developed for healthcare and other data-intensive sectors, it exemplifies the kind of cutting-edge digital infrastructure that could be adapted for biomanufacturing to create shared intelligence across distributed production sites.
Together, these pillars provide SMEs with critical infrastructure access and regulators with real-world environments for adaptive oversight. A federated network of 4–6 hubs of excellence and 12–20 specialised sites, connected through a digital backbone and governed through public–private partnerships, would operationalise this capacity under the EU Biotech Act.
Biosecurity-by-design through technology infrastructures enable the design and embedding biosecurity controls during process development, not as post-deployment retrofits. As AI-driven biotechnology becomes more accessible, governance must evolve alongside technological spread[62]. TIs can allow developers to integrate access controls, bioaudit protocols, and cyber-biosecurity measures whilst optimising manufacturing processes. Regulatory sandboxes within TIs test biosecurity frameworks in parallel with technologies, generating standards evidence. This encompasses trusted reference datasets for AI validation, hardware verification programmes, and information hazard models assessing dual-use risks. Shared infrastructures ensure next-generation biomanufacturing has built-in security from inception, ensuring the safeguarding of this part of the European industrial base.
Co-locating IMP production with clinical trial sites strengthens Europe’s capacity to retain clinical trials and accelerate development timelines. The European Federation of Pharmaceutical Industries and Associations (EFPIA) has highlighted that early-stage manufacturing infrastructure plays a decisive role in clinical trial location decisions, as proximity of IMP production reduces logistical complexity, regulatory delays, and costs. Embedding modular pilot and demonstration facilities within European clinical and regulatory ecosystems could anchor more clinical trials domestically, reinforcing Europe’s position as a preferred global site for early-stage development.
A pan-European network of interconnected hubs and sites
TIs provide the practical foundation for translating Europe’s scientific excellence into industrial capability. For SMEs, they offer access to facilities, expertise, and regulatory mechanisms that would otherwise be beyond reach – allowing early innovators to focus resources on development rather than capital investment and manufacturing scale-up, and accelerating progression to first-in-human and proof-of-concept stages. For regulators, they create structured, auditable environments in which new oversight tools and standards can be tested collaboratively, driving harmonisation and regulatory agility across Member States.
Unlike the broad, bureaucratically layered European Digital Innovation Hub (EDIH) model focused on cross-sector digital enablement, the proposed biomanufacturing network is sector-specific and regulation-integrated – meaning it embeds regulatory pathways, sandboxes, and qualification mechanisms directly into shared infrastructures – and should remain lean and mission-driven to avoid the administrative and coordination bottlenecks that challenge EDIH rollout.
Together, these pillars provide SMEs with access to critical infrastructure and regulators with real-world environments for adaptive oversight. A federated network of 4–6 hubs of excellence and 12–20 specialised sites, connected through a digital backbone and governed through public–private partnerships, would operationalise this capacity under the EU Biotech Act.
Case study
Strategic infrastructure without strategic commitment
The Vaccine Manufacturing and Innovation Centre (VMIC) illustrates both the strategic rationale for shared infrastructures and the challenges facing bold, forward-looking industrial innovation policy. VMIC emerged from post-Ebola evaluations identifying a gap in the UK’s late-stage vaccine development capacity. It was conceived as a non-profit facility supporting SMEs and academic researchers aiming to provide pilot capacity for vaccines and limited outbreak response. In May 2020, the UK Government committed €106 million to accelerate construction; total announced funding reached €250 million by March 2021[65]. Despite this, VMIC which was initiated in 2018 was sold just 4 years later in 2022 before completion[66]. The new owner (Catalent) later pledged up to €150 million to complete the facility, yet there is no evidence that surge manufacturing capacity or public-interest innovation functions were retained[67].
The VMIC case illustrates both the strategic value of shared infrastructures and the vulnerability of long new-facility projects, which can stall, be halted, or be sold off as political and funding priorities shift – its eventual sale reflected short-term fiscal pressures overriding long-term industrial strategy[68],[69]. This mirrors broader political hesitancy: a 2023 European Commission/HERA Study on flexible EU manufacturing and innovation for MCMs concludes that most publicly initiated facilities have ultimately been divested, recommending reactive tools such as CDMO contracts and capacity reservations over direct investment[70]. This contrasts with the U.S. Centers for Innovation in Advanced Development and Manufacturing (CIADM) model, which established a public–private infrastructure strategy oriented to surge production and end-to-end tech transfer well before the pandemic, enabling rapid mobilisation during COVID-19.
In the UK, the recently announced RNA Biofoundry in Darlington, co-developed with the Centre for Process Innovation (CPI), illustrates a more dynamic model of cutting-edge biomanufacturing infrastructure – anchored in public funding yet designed to integrate industry expertise and enable future private participation[71]. CPI, a not-for-profit translational research organisation, provides the technical and operational foundation for a facility capable of producing RNA materials rapidly and pivoting to vaccine manufacturing when required. Through its investment arm, CPI connects venture and industrial partners to support SME-led biomanufacturing scale-up, making this an interesting model for aligning public-purpose infrastructure with embedded expertise in leveraging private capital[72].
While fiscally cautious, the stance set out in the European Commission’s study on flexible EU manufacturing for MCMs reflects a procurement mindset [73]. To build technological sovereignty, Europe should boldly move beyond emergency tools like EU FAB and embed long-term public–private biomanufacturing infrastructure strategy within the Biotech Act to support critical scale-up stages and enable biomanufacturing and regulatory innovation.
Section 3. Biomanufacturing regulatory sandboxes de-risking novel platforms
Regulatory sandboxes give regulators and innovators the chance to step into real-world settings, test emerging technologies, observe what happens, and rethink how we regulate. Instead of crafting frameworks and guidelines in the abstract, sandboxes enable controlled experimentation in real-world conditions, revealing how, for example, automation, AI-driven manufacturing or advanced bioprocessing perform, and what unforeseen risks might arise. It’s regulation-as-lab that thrives on dialogue, expertise-sharing, and the scientific method: design, build, test.
The European Medicines Agency and the forthcoming reform of the pharmaceutical legislation have begun to explore piloting this approach in medicines development. This signals an important shift in regulatory practice – but the opportunity reaches far beyond.
Why codify regulatory sandboxes in the Biotech Act?
Embedding sandboxes into the Biotech Act would:
- Connect the pioneering work by EMA and the upcoming pharma package with the broader ambition for biotechnology and manufacturing.
- Enable regulation to grow in step with technology rather than trailing behind it.
- Break regulatory inertia by turning regulators, innovators and agencies into co-learners rather than counterparts.
- Accelerate responsible innovation by reducing uncertainty and building confidence for both developers and regulators.
In doing so, the Biotech Act would transform sandboxing from a contained pilot into a European asset of regulatory agility – a space where science, governance and industry evolve together.
In practice, regulatory sandboxes allow manufacturers, infrastructure operators and regulators to trial new production platforms in controlled conditions – independent from a product dossier[74]. Comparable approaches exist in other sectors — for instance, the UK’s MHRA AI and Software as a Medical Device Sandbox allows developers to test algorithmic decision processes, data interoperability, and validation workflows with regulators in controlled conditions before formal approval[75].
Coordination by the European Regulatory Innovation Centre (ERIC)
Embedding sandboxes into the Biotech Act would benefit from a coordinated governance mechanism to ensure coherence, foresight, and evidence-sharing across sectors. A European Regulatory Innovation Centre (ERIC) could fulfil this role by aligning sandbox initiatives under a shared EU framework for regulatory innovation (for more details see 6. Recommendations). By linking the European Commission’s policy work with the expertise of agencies such as EMA, EFSA, and HERA, ERIC would help consolidate lessons learned, avoid duplication, and accelerate the translation of experimental regulatory insights into operational guidance.
Regulatory sandboxes to break the biomanufacturing deadlock
Across major regulatory jurisdictions, there is currently no mechanism to assess and qualify biomanufacturing technologies independently of a specific product. Regulatory evaluation remains embedded within the marketing authorization process, meaning that any novel manufacturing platform introduced during development transfers regulatory risk to the entire product[76].
For large pharmaceutical companies, this creates a strong disincentive to adopt unproven biomanufacturing technologies in late-stage programmes, where regulatory delays could jeopardise multi-million- or billion-euro commercialisation strategies. Consequently, firms rely on established methods and focus on incremental process improvements. In parallel, SMEs and academic groups, often the originators of disruptive technologies, lack the capital, regulatory leverage, and infrastructure to qualify new platforms through product-specific submissions. This structural misalignment traps breakthrough biomanufacturing technologies at the pre-commercial stage.
Regulatory sandboxes can break this deadlock, allowing platform qualification and regulatory learning to take place collectively and upstream rather than product by product. It opens space for disruptive innovation, allowing new methods to move from concept to scalable reality more quickly and safely[77].
Proposal 1
Regulatory sandbox for AI-orchestrated cell-free protein synthesis
AI-Orchestrated Cell-Free Protein Synthesis (AI-CFPS) combines cell-free protein expression systems with artificial intelligence to enable distributed, flexible production of biologics. Using standardised cell extracts and AI “compiler” models, it can generate complex proteins rapidly across multiple small-scale sites, reducing dependence on centralised manufacturing and strengthening supply resilience.
Existing regulatory frameworks, designed for static, site-bound processes, are not equipped to manage this distributed and digitally orchestrated model. A regulatory sandbox could provide a controlled environment for early testing and qualification of the AI-CFPS platform under real-world conditions. It would focus on quality assurance, AI lifecycle governance, digital traceability, and data integrity, while embedding biosecurity and cyber-biosecurity-by-design. The sandbox would also clarify regulatory boundaries between the platform and its products, define what constitutes a “batch” in decentralised production, and delineate responsibilities across platform developers, AI orchestration layers, and local production sites. These activities would align with EU frameworks on responsible AI, dual-use governance, and cybersecurity resilience.
Within this framework, regulators could test tailored instruments such as:
- Platform master files documenting cell-extract validation and orchestration rules;
- Risk-based assurance models distinguishing platform-level control from product-specific oversight;
- AI governance structures consistent with GMP Annex 11 and the EU AI Act;
- Digital recipe passports linking compiler versions, production sites, and sequence data to ensure full traceability;
- Embedded biosecurity standards to promote international interoperability.
Under structured supervision, limited procedural flexibilities—such as real-time release supported by validated analytics – could be granted to assess reproducibility, reliability, and regulatory usability. Continuous AI analytics would enable predictive quality control, anomaly detection, and automated documentation, supporting real-time regulatory oversight in place of static inspection.
Embedding the sandbox within Europe’s technology infrastructures—such as distributed manufacturing networks, regulatory data spaces, and platform manufacturing registries—would transform these systems into regulatory-manufacturing innovation sites. This integration would allow real-time testing of traceability tools, master files, and analytics pipelines under operational conditions, ensuring scalability and long-term policy coherence.
An AI-CFPS regulatory sandbox would thus pioneer platform-level regulation for distributed, AI-enabled biomanufacturing and lay the foundation for future autonomous biofoundries. By combining continuous analytics, digital traceability, and platform master files within European infrastructures, it would provide the regulatory tools necessary to support rapid, flexible biomanufacturing across the Union, reinforcing Europe’s strategic autonomy in critical medical countermeasures.
Proposal 2
Regulatory sandbox for AI-enabled quality assurance for emergency use
Decentralised production of medical countermeasures (MCMs) is becoming central to Europe’s health security and biodefence preparedness. Traditional GMP systems, designed for stable and centralised production, cannot ensure the quality of emergency-use biologics made under hospital exemptions. Products such as bacteriophage cocktails are inherently adaptive, evolving with resistant pathogens and tailored to individual patients, exposing critical gaps in regulatory oversight and delaying access during crises.
AI-enabled quality assurance offers a solution for harmonised, real-time oversight across decentralised sites. Continuous process analytics, automated documentation, and deviation detection can deliver assurance equal to or greater than conventional methods, while preserving flexibility for rapid, adaptive production.
A regulatory sandbox under the Biotech Act would provide a controlled environment to test this model under real hospital exemption conditions. Bringing together national authorities, regulatory authorities, hospital manufacturers, biomanufacturing innovators and AI developers, it would evaluate AI-driven QA in practice – initially focusing on phage cocktails as a complex, high-value MCM use case. The sandbox would also demonstrate how decentralised AI systems can coordinate crisis-time manufacturing – adjusting formulations, batches, and schedules in response to infectious outbreaks, radiological incidents, or chemical exposures to maintain supply continuity under constrained conditions.
In parallel, the sandbox would assess AI’s capacity to automate regulatory data integration by generating standardised digital batch records, deviation reports, and release documentation in real time. This would streamline compliance, enable continuous regulatory oversight, and provide EU-level situational awareness during emergencies.
Findings would inform the development of EU standards for AI-based oversight and extend to decentralised biomanufacturing models generally and ones critical for biodefence, including patient-specific antimicrobial and antiviral formulations, short-lived radiopharmaceuticals for rapid diagnostics, on-demand vaccines or neutralising biologics, and 3D-printed implants for trauma care.
By embedding decentralised AI quality assurance into Europe’s regulatory framework, the sandbox would shift biodefence manufacturing from reactive to anticipatory – enabling secure, adaptive, and high-fidelity production of critical countermeasures even in high-pressure or disrupted environments.
Section 4: Biosecurity-by-design and cyber-biosecurity for next-generation biomanufacturing
Ensuring the safety, integrity, and trustworthiness of Europe’s next-generation, AI-enabled biomanufacturing ecosystem requires regulatory innovation that addresses both biological and digital dimensions of risk. Here we outline a dual framework for European biosecurity resilience and EU global leadership in standard-setting.
European and global framework for biosecurity-by-design and cyber-biosecurity in bio-digital manufacturing
As biomanufacturing moves from digitalisation to full bio-digital integration, reliant on AI-driven design, cloud-linked control and distributed production networks, Europe faces new opportunities and risks. While existing biosafety and biosecurity regulation governs biological hazards, and the Directive (EU) 2022/2555 – NIS2 strengthens cybersecurity across critical sectors in the EU, convergence of digital and biological systems exposes novel threats: cyber-intrusion, data tampering and misuse. To safeguard investment into the sector, the EU should embed biosecurity-by-design and cyber-biosecurity standards into regulatory and infrastructure frameworks, treating secure digital–biological interfaces as core quality and governance requirements.
Pillar 1: a European framework for biosecurity-by-design and cyber-biosecurity
The EU Biotech Act should establish an integrated framework embedding security and resilience across all stages of AI-enabled and digitally interconnected biomanufacturing. This framework could include:
- Integrated bio-cyber risk assessment, combining biosafety, biosecurity, and cybersecurity requirements for AI-orchestrated or autonomous biomanufacturing systems.
- Biosecurity-by-design provisions in EU funding, licensing, and certification programmes, requiring early threat modelling, access control, and human-in-the-loop safeguards for high-risk applications.
- Alignment with NIS2, the AI Act, and GMP Annex 11, extending cybersecurity and AI governance principles to biotechnology infrastructures.
- Anticipate alignment with forthcoming EU Cloud and AI Development Acts , to ensure coherent internal-market standards for bio-digital infrastructures.
- A European Cyber-Biosecurity Coordination Mechanism linking ENISA, EMA, HERA, and DG HOME to oversee standards development, incident reporting, and capacity building.
This approach would ensure that Europe’s biotechnology ecosystem operates under coherent, risk-proportionate safeguards while maintaining innovation agility and competitiveness.
Pillar 2: global leadership and standard-setting
In parallel, the EU should leverage its normative and regulatory influence to promote international convergence through a Global Standards and Governance Framework for Secure Bio-Digital Manufacturing, including:
- Cooperation with key organisations such as the OECD, WHO, and the Australia Group to develop shared biosecurity-by-design and cyber-biosecurity principles.
- Harmonisation of EU and global standards to enable interoperability and mutual recognition of secure biomanufacturing practices.
- Development of reference architectures and certification schemes for trusted AI-enabled biomanufacturing.
- Promote the establishment of consistent, globally-recognised screening standards for DNA synthesis and compiler systems, and encourage continual innovation in screening practices so they can keep pace with rapidly evolving synthesis technologies.
- Use of EU diplomatic and trade instruments—including the EU–U.S. Trade and Technology Council—to promote global convergence on responsible and secure bio-digital innovation.
A harmonised European and global framework would protect Europe’s bio-digital infrastructure, bolster its resilience and strategic autonomy, and lay the regulatory groundwork for the most advanced innovations.
Biodefence in a cloud-driven, bio-digital future
Picture this scenario: during a post-authorisation vaccine study, a European manufacturer using an AI-assisted, cloud-connected production system became the target of a coordinated digital attack, allegedly linked to a rival pharmaceutical company. The attackers exploited weaknesses in the AI-controlled bioprocess systems, subtly altering production conditions in a way that affected the consistency of vaccine batches supplied to trial sites.
Bio-Digital Threat Dynamics
The attackers exploited the algorithmic control layer of the production process, manipulating AI calibration and optimisation parameters governing temperature, pressure, and nutrient flows. These subtle algorithm changes, imperceptible to human supervisors, disrupted batch uniformity and reduced vaccine potency, leading to inconsistent immunogenicity across trial participants and sabotaging trial success.
While analogous sabotage could theoretically occur in conventional biomanufacturing, AI integration significantly amplifies the attack surface:
- Automation Dependency: In AI-managed systems, deviations can propagate at scale before human detection, whereas manual systems rely on more frequent physical checks.
- Data-Model Feedback Loops: Attacks that manipulate training or calibration data can alter system behaviour persistently, extending impact beyond a single event.
- Cloud Connectivity: Continuous remote access and data integration create new vectors for cross-domain interference between cyber and biological domains.
Implications for Biodefence and Market Stability
This type of interference could derail a pivotal clinical study, erode confidence in authorised vaccines, and damage Europe’s capacity to ensure secure, trusted biotechnological innovation. The hypothetical incident underscores the growing intersection between biotechnology and digital systems – and the amplified vulnerabilities that accompany AI-enabled automation in biotechnology manufacturing.
Section 5. Legislative tools to bridge the second valley of death
Move biotechnology from the lab to the factory to the market
The forthcoming EU Biotech Act seeks to establish a coherent framework for the development, scale-up, and deployment of biotechnologies across the Union. This initiative forms part of the Union’s broader ambition to reinforce technological sovereignty by enabling biotechnology to move, in the words of President Ursula von der Leyen, “from the laboratory to the factory and then onto the market”.
It has been suggested that the legislative scope for the EU Biotech Act I will be Articles 114 and 179 TFEU, and for the EU Biotech Act II, Article 114 TFEU [78]. These provisions each address distinct but complementary policy domains:
- Article 114 TFEU enables the harmonisation of national rules that affect the placing on the market of products or services, such as medicinal products, chemicals, or medical devices. It regulates market-entry conditions, safety standards, and post-authorisation requirements, but does not cover pre-commercial industrial development. Its relevance arises only once a technology or product is sufficiently mature to enter the internal market.
- Article 179 TFEU establishes the Union’s competence to “strengthen its scientific and technological bases” and to “encourage undertakings, including SMEs, research centres, and universities in their research and technological development activities.” It underpins the Horizon Europe framework and other research programmes focused on R&D and international cooperation. Its scope is limited to research and knowledge generation, enabling research infrastructures but not production, pilot, or demonstration facilities.
Together, Articles 114 and 179 provide a sound basis for advancing research and regulatory harmonisation, yet they do not encompass the industrial deployment phase – the translation of research outcomes into scalable, competitive production. This is precisely where the Union faces the greatest structural gap, often described as the “second valley of death”.
A tripartite legal architecture for innovation-to-market coverage
To bridge this gap and deliver on the Commission’s stated ambition of bringing biotechnology “to the factory and then onto the market” Article 173 TFEU should be introduced as a third, complementary legal foundation. It empowers the Union and the Member States to “ensure that the conditions necessary for the competitiveness of the Union’s industry exist” and to “foster better exploitation of the industrial potential of policies of innovation, research and technological development”. This article provides the necessary legal underpinning for technology infrastructures – including shared pilot lines, pilot plants, and demonstration facilities – that enable industrial scaling, regulatory learning, and biomanufacturing innovation within the Union.
The inclusion of Article 173 TFEU establishes a tripartite legal structure that ensures coverage across the full innovation-to-market continuum:
Article 179 TFEU – governs the research and innovation phase, supporting scientific excellence, R&D collaboration, and research infrastructures.
Article 173 TFEU – governs the industrial deployment phase, enabling scale-up, pilot manufacturing, and technology infrastructures that convert innovation into production capacity.
Article 114 TFEU – governs the market phase, harmonising product rules, safety standards, and authorisation conditions across the internal market.
Establishing a coherent legal base for strategic technology competitiveness
Across the Union’s emerging high-technology legislative framework, Article 173 TFEU has become a consistent legal basis for supporting the scaling, coordination, and market-enabling conditions necessary to develop Europe’s industrial capabilities in strategic technologies. The Chips Act, Quantum Act, Advanced Materials Initiative, and European Innovation Act all plan to integrate Article 173, alongside Articles 114, 179, or related provisions, to link research, industrial deployment, and market integration within a coherent strategic framework[79]:
Chips Act
173,144
Pilot lines and semiconductor manufacturing capacity
Quantum Act (planned)
173, 180, 184
Industrial ecosystems and research infrastructures
Advanced Materials Initiative
173, 114
Industrial scaling and test-bed infrastructures
European Innovation Act (planned)
114, 173, 182
Innovation infrastructures and technology uptake
Embedding Article 173 TFEU in the EU Biotech Act would ensure alignment with this legislative pattern, reinforcing coherence across the Union’s high-technology initiatives and positioning biotechnology within the same industrial and technological governance model that underpins Europe’s broader competitiveness agenda.
The application of Article 173 is inherently shaped by the principle of subsidiarity: the Union may act where collective investment and coordination achieve results that Member States cannot attain effectively alone. In this context, EU action should focus on establishing cross-border networks of technology infrastructures, co-financed through Union and national instruments, while Member States retain responsibility for implementation and ownership. This framework allows the Union to act as a catalyst – aligning priorities, pooling resources, and ensuring coherence across national initiatives – while respecting national industrial prerogatives and diversity.
Section 6. Key recommendations for the EU Biotech Act
1. Technology infrastructures for scaling innovation to market
A sector-specific TI framework, embedded within the Biotech Act and aligned with the European Strategy on Research and Technology Infrastructures, should include:
- Biofoundries to accelerate design-to-manufacture translation and process optimisation;
- Modular pilot and demonstration facilities to de-risk scale-up and anchor early clinical manufacturing in Europe;
- Regulatory sandboxes to promote regulatory learning and adaptive oversight through real-world technology qualification with EU and national authorities;
- Federated digital data environments to build biomanufacturing collective intelligence, ensuring interoperability, traceability, and biosecurity-by-design principles that set European standards.
A pan-European network of 4–6 hubs of excellence and 12–20 specialised sites, governed through public–private partnerships and SME access mechanisms, would operationalise Europe’s infrastructure strategy, accelerate biomanufacturing scale-up, and reinforce the Union’s global technological leadership. [more details in 2. Technology Infrastructure]
2. Modernise the EU regulatory framework for biomanufacturing across biotechnology sectors
The EU Biotech Act should modernise, align and future-proof regulatory frameworks to reflect the central role of biomanufacturing as a cross-cutting capability underpinning Europe’s biotechnology sectors. Regulation should evolve from product-specific oversight to platform-based, adaptive, and digitally integrated approaches, ensuring both innovation speed and public trust.
Key priorities:
- Establish Platform Technology Master Files (PTMFs) to enable one-time assessment of core biomanufacturing systems, reducing duplication across applications.
- Harmonise contained-use and GMO procedures to create consistent, proportionate rules across Member States.
- Extend adaptive qualification and real-world evidence (RWE) approaches to biomanufacturing systems, enabling regulators to authorise core platforms while accommodating rapid updates and continuous improvements and enabling distributed and continuous production models.
- Integrate AI lifecycle management and digital traceability frameworks to document training data, compiler versions, site, and batch information, ensuring transparency, comparability, and rapid oversight across decentralised networks.
- Embed biosecurity-by-design and cyber-biosecurity within platform-level certification to secure bio-digital systems.
- Prioritise regulatory training and investment under Horizon Europe, EU4Health, and Digital Europe to strengthen scientific and digital expertise, and ensure coherent oversight across health, industrial, and digital domains.
This would update a coherent, forward-looking regulatory framework that enables biomanufacturing to drive innovation, resilience, and competitiveness across Europe’s entire biotechnology landscape. [more details in 1. Biomanufacturing innovation & regulatory reform]
3. Establish a European Regulatory Innovation Centre (ERIC)
A European Regulatory Innovation Centre (ERIC) should be established to coordinate foresight, evidence generation, and regulatory innovation across deep tech sectors, including biotechnology.
ERIC should serve as a permanent coordination and foresight body supporting both the European Commission’s policy Directorates-General (DGs) and the Joint Research Centre (JRC), while working in close cooperation with EU regulatory agencies such as EMA, EFSA, ECHA, and HERA.
Its core functions should include:
- Developing EU-wide frameworks and standards for foresight, ensuring methodological coherence and interoperability across Commission DGs, the JRC, and agencies, while enabling cross-regulatory analysis of deep tech interfaces.
- Commissioning and synthesising targeted research on regulatory innovation, including risk–benefit analysis, socio-economic impact, standards mapping, and safety-by-design approaches relevant to converging technologies.
- Maintaining an open EU Evidence Repository to consolidate foresight outputs, research findings, and regulatory intelligence, ensuring transparent access for agencies, policymakers, and Member States.
- Coordinating and developing regulatory innovation tools, such as sandboxes, adaptive guidance mechanisms, and test beds, with a focus on cross-sector applicability and shared learning.
- Translating foresight and research into implementation guidance, bridging the gap between conceptual policy design and operational regulatory execution across sectors.
- Monitoring regulatory performance and identifying emerging overlaps or barriers, providing evidence-based recommendations on regulatory coherence and modernization priorities.
By consolidating currently fragmented foresight and regulatory innovation activities, ERIC would create a strategic, evidence-based, and forward-looking regulatory ecosystem capable of managing the intersecting regulatory dimensions of deep tech, ensuring agile, coherent, and science-driven governance across biotechnology and other transformative sectors in the European Union.
4. Qualification and accelerated pathways for disruptive biomanufacturing innovation
Under current rules, novel manufacturing processes can only be introduced as part of full product marketing authorisations, which discourages risk-taking and slows uptake. The EU Biotech Act should:
- Establish a Biomanufacturing Innovation Qualification (BIQ) pathway to assess and approve platform and process innovations independently of specific products.
- Develop EU comparability frameworks for analytics, stability, and platform switching.
- Introduce an accelerated qualification pathway for truly cross-cutting, disruptive, high-impact biomanufacturing innovations – those that demonstrably advance EU resilience, health security, climate action, or sustainability – for the benefit of citizens.
- Integrate BIQ within a cross-agency coordination framework linking the EMA, HERA, EFSA, ECHA, and national authorities, ensuring coherent qualification, preparedness, and technology transfer across sectors by embracing adaptive qualification mechanisms.
- Provide targeted regulatory and funding support to first movers to encourage early adoption.
This would remove the “innovation penalty” currently associated with integrating advanced biomanufacturing technologies into product pipelines.
5. AI-enabled regulatory assistant for developers
Develop a publicly available AI Regulatory Assistant to help developers, particularly SMEs small navigate complex and overlapping EU biotechnology regulations. Core Actions include:
- Train the system on EU and national regulatory texts, prior agency opinions, and case precedents.
- Provide interactive decision-support on product classification, pathway selection, and dossier preparation.
- Integrate explainable AI to ensure transparency and trust in regulatory guidance.
- Maintain continuous updates through collaboration with ERIC and EU regulatory agencies.
This would accelerate translation of innovation to market through improved regulatory clarity, reduced administrative burden, and faster alignment between developers and authorities.
6. Establish a European framework for biosecurity-by-design and cyber-biosecurity
The EU Biotech Act should embed biosecurity-by-design and cyber-biosecurity principles across regulatory, funding, and certification frameworks to ensure safe, trustworthy, and resilient AI-enabled biomanufacturing. Core actions include:
- Integrate biosecurity and biosafety risk assessment early in process design, technology qualification, and facility certification.
- Require threat modelling, access control, and human-in-the-loop safeguards for high-risk biomanufacturing applications.
- Develop a coordinated European cyber-biosecurity framework linking ENISA, EMA, HERA, and DG HOME to secure bio-digital infrastructures and enable integrated risk assessment and incident reporting.
- Align provisions with GMP Annex 11, the AI Act, and NIS2, ensuring consistent digital–biological quality and safety standards.
- Promote international standard-setting with OECD, WHO, and the Australia Group to advance reference architectures, certification schemes, and global convergence on secure bio-digital innovation.
This would ensure a harmonised European and international framework that embeds security throughout biomanufacturing design and governance – safeguarding bio-digital infrastructure, reinforcing EU resilience, and positioning Europe as a global leader in secure biotechnology. [more details in 4. Biosecurity]
7. Base the EU Biotech Act on a tripartite legal framework encompassing the full innovation-to-market continuum
The EU Biotech Act should be grounded in a tripartite legal framework combining Articles 179, 173, and 114 TFEU, reflecting the structure of other strategic technology initiatives such as the Chips Act and the Advanced Materials Initiative. Core actions include:
- Use Article 179 TFEU for research and innovation; Article 173 TFEU for industrial deployment and technology infrastructures; and Article 114 TFEU for market integration and product harmonisation.
- Ensure coherence with the Union’s broader framework for strategic technologies and industrial competitiveness.
- Focus EU action on cross-border coordination, shared pilot facilities, and co-investment mechanisms, while respecting Member State implementation responsibilities.
This would ensure a coherent legal foundation enabling biotechnology to move from research to industrial scale and market uptake—bridging Europe’s second valley of death and aligning the sector with the Union’s wider high-technology governance model. [more details in 4. Biosecurity]
Annex
Shaping the EU Biotech Act: insights from SME and cluster Representatives
Briefing note on the discussions
About this initiative
The upcoming EU Biotech Act represents a major opportunity to shape Europe’s competitiveness in biotechnology and biomanufacturing. The Act aims to create a coherent, cross-sector framework to accelerate innovation, strengthen regional bioeconomies, and ensure that Europe remains a global leader in health and life sciences.
However, small and medium-sized biotech enterprises (SMEs), which drive much of the innovation pipeline, remain underrepresented in current policy debates. Most lack dedicated teams or structured channels to inform EU-level policymaking.
To address this gap, Centre for Future Generations (CFG), together with Amgen, co-hosted a workshop in 2025 bringing together biotech clusters and SME representatives from across Europe. The goal was to gather concrete input on the challenges SMEs face in manufacturing, regulatory navigation, intellectual property (IP), and market access, and to formulate actionable insights for EU policymakers shaping the Biotech Act.
The session included two guided breakout discussions:
- Breakout 1: Manufacturing and Supply Chain
- Breakout 2: Regulatory Navigation, IP and Market Access
The workshop brought together six participants representing key parts of Europe’s health biotechnology ecosystem, including two regional innovation clusters, one national biotech association, and two EU-level trade and policy organisations. Collectively, these organisations represent over 400 small and medium-sized biotech companies across Europe, spanning early-stage start-ups to clinical development SMEs, with a combined annual turnover estimated at over €2 billion.
*Please note: the views below do not represent the CFG’s or Amgen’s perspectives. This briefing provides an overview of the discussions held by the participants.
Summary of discussions
Across both breakout discussions, participants highlighted that Europe’s biotechnology ecosystem is rich in scientific talent but constrained by structural barriers, preventing SMEs from progressing from discovery to market.
Five recurring themes emerged:
1. Manufacturing and scale-up bottlenecks
SMEs struggle to access small-scale GMP manufacturing for clinical trials, with limited pilot infrastructure and long lead times. Shared facilities and targeted EU programmes that improve access to manufacturing infrastructure were seen as essential to enable SMEs to translate innovation into viable therapies.
2. Funding and regional imbalances
Funding gaps persist at both EU and national levels, particularly for manufacturing and scale-up process innovation, digitalisation, and AI integration. EU structural funds often exclude innovation hubs located in “wealthier” regions, decoupling support from actual biotech activity.
3. Regulatory fragmentation and resource strain
The EU regulatory environment remains fragmented, with limited access to timely, biotech-specific advice. Participants called for stronger EMA capacity, better coordination between regulatory and HTA bodies, and digital “one-stop” support for SMEs.
4. Skills and workforce shortages
Upscaling from laboratory to GMP production requires a specialised skill set rarely developed in universities and independent quality oversight. Public–private training partnerships, modelled on existing cluster-led programmes, were seen as critical to closing this gap.
5. Intellectual property and market access predictability
SMEs face high costs and inconsistent guidance when seeking IP protection. Clear, affordable, and reliable EU-level advisory support is needed to avoid early strategic missteps. Lengthy national reimbursement processes also erode the value of exclusivity, underscoring the need for mechanisms that safeguard the effective duration of innovation incentives.
The calls to action
- Create shared small-scale GMP facilities and pilot plants accessible across Member States, ensuring equitable access for SMEs developing innovative modalities such as gene and cell therapies following appropriate GMPs.
- Reform EU funding eligibility to link grant support to innovation activity rather than Cohesion Policy funding based on regional GDP classification , enabling facilities to be built where biotech companies actually operate.
- Establish an EU-level “Biotech One-Stop Hub” combining scientific, regulatory, and manufacturing guidance, supported by EMA and DG SANTE. This hub should also offer coordinated advice for combination and platform-based products to reduce duplication and uncertainty.
- Introduce proportional regulatory pathways for SMEs, allowing adaptive approaches and sandbox models for emerging technologies.
- Advance regulatory harmonisation and mutual recognition across Member States for biotech manufacturing, clinical trials, and product evaluation. Greater alignment of assessment procedures and recognition of equivalent quality standards would shorten approval timelines and lower compliance costs for companies across all stages.
- Launch an EU Biotech Skills Initiative that funds industry-academic partnerships to train process engineers and translational specialists.
- Align IP and market access frameworks by clarifying patentability for AI-driven discoveries, harmonising evidence expectations across regulatory and HTA bodies, and compensating for lost exclusivity time due to pricing delays.
- Integrate the Biotech Act with funding instruments (e.g. IPCEIs, Horizon Europe) to ensure coherence between policy goals and investment mechanisms.
Full summary of workshop discussions
Breakout 1: Manufacturing and supply chain
Breakout 2: Regulatory navigation, IP, and market access
Endnotes
[1]European Commission, Health Emergency, Preparedness and Response Authority (HERA), 2023 Annual Work Plan of the Health Emergency, Preparedness and Response Authority (HERA), annex (Brussels: European Commission, 2023) health.ec.europa.eu.
[2]European Commission, EU strategic dependencies and capacities: second stage of in‑depth reviews, Commission Staff Working Document SWD(2022) 41 final (Brussels, 22 February 2022) wec-italia.org.
[3]NVIDIA ‘Lilly Deploys World’s Largest, Most Powerful AI Factory for Drug Discovery Using NVIDIA Blackwell-Based DGX SuperPOD’, NVIDIA Blog. (2025) https://blogs.nvidia.com/blog/lilly-ai-factory-nvidia-blackwell-dgx-superpod/
[4] BioVox (2016) ‘From lab to market: Pilot plants are key’.
[5]François Candelon, Matthieu Gombeaud, Georgie Stokol, Vinit Patel, Antoine Gourévitch and Nicolas Goeldel, ‘Synthetic biology is about to disrupt your industry’, Boston Consulting Group (10 Feb 2022) https://www.bcg.com/publications/2022/synthetic-biology-is-about-to-disrupt-your-industry
[6]European Medicines Agency (EMA), ‘Development of a guideline on the quality aspects of mRNA vaccines – scientific guideline’, reference EMA/CHMP/BWP/82416/2025 (first published 31 Mar 2025) https://www.ema.europa.eu/en/development-guideline-quality-aspects-mrna-vaccines-scientific-guideline
[7]European Directorate for the Quality of Medicines & HealthCare (EDQM), ‘European Pharmacopoeia Commission adopts first three general texts on mRNA vaccines’, Newsroom, Strasbourg, 7 Jan 2025 https://www.edqm.eu/en/-/european-pharmacopoeia-commission-adopts-first-three-general-texts-on-mrna-vaccines
[8]John H. Skerritt, ‘Considerations for mRNA product development, regulation and deployment across the lifecycle’, Vaccines, 13(5) (2025), article 473 https://www.mdpi.com/2076-393X/13/5/473
[10]L. Sundaram, J. W. Ajioka and J. C. Molloy, ‘Synthetic biology regulation in Europe: containment, release and beyond’, Synthetic Biology, vol. 8, no. 1 (20 Apr. 2023)scispace.com.
[11]European Commission, ‘Genetically modified organism (GMO) aspects for investigational medicinal products’, Public Health (n.d.)health.ec.europa.eu.
[12]Boyds Consultants, Clinical Trials with Genetically Modified Organisms (EU), Regulatory Factsheet 6 (2024)boydconsultants.com.
[13]DLRC Group, ‘Platform technology: how to reference prior knowledge’, 31 Mar. 2025dlrcgroup.com.
[14]Michael L. King, ‘How manufacturing won or lost the COVID‑19 vaccine race’, Vaccine, 42(5) (Feb 2024), 1004–1012 https://www.sciencedirect.com/science/article/pii/S0264410X23014779#b0300
[15]J. F. Zawada et al., ‘Microscale to manufacturing scale‑up of cell‑free cytokine production—a new approach for shortening protein production development timelines’, Biotechnology and Bioengineering, vol. 108, no. 7 (July 2011), 1570–1578pubmed.ncbi.nlm.nih.gov
[16]K. Khambhati et al., ‘Exploring the potential of cell‑free protein synthesis for extending the abilities of biological systems’, Frontiers in Bioengineering and Biotechnology, vol. 7 (11 Oct. 2019), article 248pubmed.ncbi.nlm.nih.gov.
[17]J. F. Zawada et al., ‘Cell‑free technologies for biopharmaceutical research and production’, Current Opinion in Biotechnology, vol. 76 (Aug. 2022), 102719pubmed.ncbi.nlm.nih.gov.
[18]O. Borkowski et al., ‘Large scale active‑learning‑guided exploration for in vitro protein production optimization’, Nature Communications, vol. 11 (20 Apr. 2020), article 1872pubmed.ncbi.nlm.nih.gov.
[19]A. D. Silverman, A. S. Karim and M. C. Jewett, ‘Cell‑free gene expression: an expanded repertoire of applications’, Nature Reviews Genetics, vol. 21, no. 3 (Mar. 2020), 151–170pubmed.ncbi.nlm.nih.gov.
[20]Ian Betteridge, ‘AI‑driven biomanufacturing: revolutionising production and quality in pharmaceuticals’, European Pharmaceutical Review, 28 Apr https://www.europeanpharmaceuticalreview.com/article/254402/ai-driven-biomanufacturing-revolutionising-production-and-quality-in-pharmaceuticals/
[21]Robert Dream, ‘The use of AI and ML in biomanufacturing: process and product validation lifecycle and AI’, American Pharmaceutical Review, 1 Feb. 2024americanpharmaceuticalreview.com.
[22]Boehringer Ingelheim, ‘Sutro Biopharma and Boehringer Ingelheim BioXcellence™ collaboration: established first‑in‑class cell‑free capabilities at commercial scale’, press release, 8 Jan. 2025pharmafocuseurope.com.
[23]M. T. Smith, S. D. Berkheimer, C. J. Werner and B. C. Bundy, ‘Lyophilized Escherichia coli‑based cell‑free systems for robust, high‑density, long‑term storage’, BioTechniques, vol. 56, no. 4 (1 Apr. 2014), 186–193pubmed.ncbi.nlm.nih.gov.
[24]Brian J. Ward et al., ‘Phase 1 randomized trial of a plant‑derived virus‑like particle vaccine for COVID‑19’, Nature Medicine, vol. 27 (June 2021), 1071–1078pubmed.ncbi.nlm.nih.gov.
[25]Somen Nandi et al., ‘Techno‑economic analysis of a transient plant‑based platform for monoclonal antibody production’, mAbs, vol. 8, no. 8 (Nov.–Dec. 2016), 1456–1466pubmed.ncbi.nlm.nih.gov.
[26]Mintu Mathew and Jaya Thomas, ‘Tobacco‑based vaccines, hopes, and concerns: a systematic review’, Molecular Biotechnology, vol. 65, no. 7 (July 2023), 1023–1051pubmed.ncbi.nlm.nih.gov.
[27]Brian J. Ward et al., ‘Phase 1 randomized trial of a plant‑derived virus‑like particle vaccine for COVID‑19’, Nature Medicine, vol. 27 (June 2021), 1071–1078pubmed.ncbi.nlm.nih.gov.
[28]Jonathan Gardner, ‘Vaccine from Medicago, GSK protective against COVID‑19 in large study’, BioPharma Dive, 7 Dec. 2021biopharmadive.com.
[29]Johannes Buyel, Josef Glössl and Eva Stöger, Using plant molecular farming to increase regional biomanufacturing capacity across the globe for fast, resilient and cost‑efficient medicine supply, Science‑Policy Brief for the UN Multistakeholder Forum on Science, Technology and Innovation (May 2024)sdgs.un.org.
[30]Stefan Schillberg and Ricarda Finnern, ‘Plant molecular farming for the production of valuable proteins – critical evaluation of achievements and future challenges’, Journal of Plant Physiology, 258–259 (Mar 2021), article 153359 https://www.sciencedirect.com/science/article/pii/S0176161720302492
[31]J. W. Huebbers and J. F. Buyel, ‘On the verge of the market – plant factories for the automated and standardised production of biopharmaceuticals’, Biotechnology Advances, vol. 46 (Jan.–Feb. 2021), 107681pubmed.ncbi.nlm.nih.gov.
[32]Juliane Steingroewer (ed.), Plants as factories for bioproduction: recent developments and applications, Advances in Biochemical Engineering/Biotechnology 188 (Cham: Springer, 2024)link.springer.com.
[33]Feliza Mirasol, ‘Progressing toward full automation in biomanufacturing’, BioPharm International, vol. 38, issue 2 (11 Mar. 2025), 6–8biopharminternational.com.
[34]Danish J. Malik et al., ‘Advanced manufacturing, formulation and microencapsulation of therapeutic phages’, Clinical Infectious Diseases, vol. 77, supplement 5 (Nov. 2023), S370–S383storre.stir.ac.uk.
[35]Elham Mohammadi et al., ‘Bridging the gap: phage manufacturing processes from laboratory to agri‑food industry’, Virus Research, vol. 353 (Mar. 2025), 199537pubmed.ncbi.nlm.nih.gov.
[36] X. Wu et al., ‘Advanced strategies in phage research: innovations, applications, and challenges’, Microorganisms, vol. 13, no. 8 (2025), article 1960mdpi.com.
[37]Silvina Alicia Pujato, Mariángeles Briggiler‑Marcó and Diego Javier Mercanti, ‘Phage biocontrol: enhancing food safety through effective pathogen reduction’, Advances in Food and Nutrition Research, vol. 116 (2025), 359–407pubmed.ncbi.nlm.nih.gov.
[38]Chinmay V. Tikhe and George Dimopoulos, ‘Phage therapy for mosquito larval control: a proof‑of‑principle study’, mBio, 13(6) (20 Dec 2022), doi:10.1128/mbio.03017‑22 https://journals.asm.org/doi/10.1128/mbio.03017-22
[39]Stephen C. Emencheta, Adaeze L. Onugwu, Chisom F. Kalu, Patience N. Ezinkwo, Osita C. Eze, Marta M. D. C. Vila, Victor M. Balcão, Anthony A. Attama and Ebele B. Onuigbo, ‘Bacteriophages as nanocarriers for targeted drug delivery and enhanced therapeutic effects’, Materials Advances, 5 (2024), 986–1016 https://www.sciencedirect.com/org/science/article/pii/S2633540924000732
[40]Hui Wang, Ying Yang, Yan Xu, Yi Chen, Wenjie Zhang, Tianqing Liu, Gang Chen and Kaikai Wang, ‘Phage‑based delivery systems: engineering, applications, and challenges in nanomedicines’, Journal of Nanobiotechnology, 22(1) (25 June 2024), 365 https://pubmed.ncbi.nlm.nih.gov/38918839/
[41]Xiaoxian Kuang, Juntao Shen, Linggang Zheng, Yi Duan, Yingfei Ma, Elaine Lai‑Han Leung and Lei Dai, ‘Applications of bacteriophages in precision engineering of the human gut microbiome’, Engineering Microbiology, 5, no. 1 (1 May 2025), article 100189 journal.hep.com.cnhttps://www.sciencedirect.com/science/article/pii/S2667370325000013
[42]Nicholas Guise, David Pattie, Kenneth B. Yeh, Kemper Talley and Rachel Fennell Fezzie, ‘2023 cyberbiosecurity summit underscores challenges associated with cybersecurity and the rapidly growing bioeconomy’, Global Security: Health, Science and Policy, 9, no. 1 (2024)
https://www.tandfonline.com/doi/full/10.1080/23779497.2024.2401164#abstract
[43]European Commission The EU Startup and Scaleup Strategy: Choose Europe to start and scale, COM(2025) 270 final. Brussels: European Commission. (PDF)
[44] Centre for Future Generations Shaping the EU Biotech Act: insights from SME and cluster representatives, unpublished (2025) annex
[45]European Commission, ‘European Chips Act’, Shaping Europe’s Digital Future (21 Sept. 2023)digital-strategy.ec.europa.eu.
[46]GFI Europe, ‘Bio Base Europe – visiting the bridge over the innovation valley of death’ https://gfieurope.org/blog/bio-base-europe-bridge-over-innovation-valley-of-death/
[47] European Commission, ‘The European Strategy on Research and Technology Infrastructures’, Research and Innovation (15 Sept. 2025)research-and-innovation.ec.europa.eu.
[48]J. Philp and D. Winickoff, ‘Innovation ecosystems in the bioeconomy’, OECD Science, Technology and Industry Policy Papers, no. 76 (OECD Publishing, 2019)oecd.org.
[49] Centre for Future Generations Shaping the EU Biotech Act: insights from SME and cluster representatives, unpublished (2025) annex
[50]Erica L. Friedman, ‘Top trends in global biomanufacturing: 20 years of analysing biologics production and supply’, BioProcess International, 22(1–2) (Jan.–Feb. 2024)eu-assets.contentstack.com.
[51]Sue Pearson: U.K. Boosts Presence at the Biologics Manufacturing Table, Genetic Engineering & Biotechnology News,(4 January 2024) https://www.genengnews.com/topics/bioprocessing/uk-boosts-presence-at-the-biologics-manufacturing-table/
[52] Singapore Economic Development Board (EDB), ‘Why Singapore is a top choice for biopharma firms seeking global expansion’, Business Insights (20 Oct. 2023)edb.gov.sg.
[53]President’s Council of Advisors on Science and Technology (PCAST), Biomanufacturing Report (Dec. 2022) https://bidenwhitehouse.archives.gov/wp-content/uploads/2022/12/PCAST_Biomanufacturing-Report_Dec2022.pdf
[54]National Security Commission on Emerging Biotechnology, ‘Charting the Future of Biotechnology: An Action Plan for American Security and Prosperity’ (April 2025) https://www.biotech.senate.gov/final-report/chapters/
[55]Pawel Swieboda, ‘Wired for the Future: Boosting Europe’s Neurotechnology Frontier’ (Centre for Future Generations, 19 September 2025) https://cfg.eu/wired-for-the-future/
[56]European Commission, Directorate‑General for Research and Innovation, User needs for technology infrastructures: Analytical report (Brussels, 2025)op.europa.eu.
[57]European Commission, ‘The European Strategy on Research and Technology Infrastructures’, Research and Innovation (15 Sept. 2025)research-and-innovation.ec.europa.eu.
[58]Emma Pirnay, ‘INTERVIEW: EU must “continue to uphold science”, says head of drug agency’, Euractiv, 20 Oct 2025 https://www.euractiv.com/interview/interview-eu-must-continue-to-uphold-science-says-head-of-drug-agency/
[59]European Innovation Council and SMEs Executive Agency, Unlocking Innovation through Corporate-Startup Collaboration: The EIC Corporate Partnership Programme (European Commission 2025) https://op.europa.eu/en/publication-detail/-/publication/018e22db-5d34-11f0-a9d0-01aa75ed71a1/language-en
[60] European Union, Policy Landscape Supporting Technology Infrastructures in Europe – Final Report (Publications Office of the European Union 2024) https://data.europa.eu/doi/10.2777/504517
[61]NVIDIA, ‘NVIDIA FLARE: Federated Learning Application Runtime Environment’, NVIDIA Developer (n.d.)developer.nvidia.com.
[62] Drew Endy et al., Biosecurity Really: A Strategy for Victory (Stanford CA: Hoover Institution, Oct. 2025)hoover.org.
[63] T. Wilsdon, H. Armstrong, A. Sablek and P. Cheng, Factors affecting the location of biopharmaceutical investments and implications for European policy priorities – Final report (London: Charles River Associates for EFPIA, 3 Oct. 2022)efpia.eu.
[64] Centre for Future Generations Shaping the EU Biotech Act: insights from SME and cluster representatives, unpublished (2025) annex
[65] UK Department of Health and Social Care, ‘Extra £47.6 million for Vaccines Manufacturing and Innovation Centre’, GOV.UK (20 Mar. 2021)gov.uk
[66] UK Parliament, House of Lords, Written Question HL6823 (Lord Bourne of Aberystwyth) to the Department for Science, Innovation and Technology about the Vaccines Manufacturing and Innovation Centre, answered 11 Apr. 2023questions-statements.parliament.uk.
[67] Catalent, ‘Catalent acquires facility in Oxfordshire to expand biologics capabilities in the UK and across Europe’, press release, 6 Apr. 2022catalent.com.
[68] R. E. Glover, A. P. Roberts, A. C. Singer and C. Kirchhelle, ‘Sale of UK’s Vaccine Manufacturing and Innovation Centre’, BMJ, vol. 376 (23 Feb. 2022), doi:10.1136/bmj‑2022‑069999bmj.com.
[69] Patrick Daly, ‘Sale of UK vaccine manufacturing plant branded “ridiculously short‑sighted”’, The Independent, 6 Apr. 2022independent.co.uk.
[70]European Union, A Study Assessing Scientific, Engineering, Legal and Economic Considerations of Flexible EU Manufacturing and Innovation of Medical Countermeasures for Serious Cross-Border Threats to Health (Publications Office of the European Union 2023) https://op.europa.eu/en/publication-detail/-/publication/cae10004-0e5b-11ee-b12e-01aa75ed71a1/language-en
[71]UK Department for Science, Innovation and Technology, ‘Next‑gen therapies for cancer, dementia and more fast‑tracked with new facility’, GOV.UK (28 Aug. 2025)gov.uk.
[72] Ian Smith, ‘Our CPI Enterprises value creation process’, CPI blog (17 Jul. 2023)uk-cpi.com.
[73]European Union, A Study Assessing Scientific, Engineering, Legal and Economic Considerations of Flexible EU Manufacturing and Innovation of Medical Countermeasures for Serious Cross-Border Threats to Health (Publications Office of the European Union 2023) https://op.europa.eu/en/publication-detail/-/publication/cae10004-0e5b-11ee-b12e-01aa75ed71a1/language-en
[74]Peter Arlett, Falk Ehmann, Ralf Herold, Evdokia Korakianiti, Pierpaolo Moscariello, Liese Barbier, Iordanis Gravanis, Frank Pétavy, Emmanuel Cormier, Andrew Thomson, Spiros Vamvakas and Steffen Thirstrup, ‘A fast path from innovation to safe and effective medicines’, Clinical Pharmacology & Therapeutics (first published online 28 Oct 2025), doi:10.1002/cpt.70107 https://ascpt.onlinelibrary.wiley.com/doi/10.1002/cpt.70107?af=R
[75]Medicines and Healthcare products Regulatory Agency (MHRA), “AI Airlock: Regulatory Sandbox for AI as a Medical Device” (UK Government, first published 2023), https://www.gov.uk/government/collections/ai-airlock-the-regulatory-sandbox-for-aiamd
[76]National Academies of Sciences, Engineering, and Medicine (2021) Innovations in Pharmaceutical Manufacturing on the Horizon: Technical Challenges, Regulatory Issues, and Recommendations. Washington, DC: The National Academies Press.https://nap.nationalacademies.org/catalog/26009/innovations-in-pharmaceutical-manufacturing-on-the-horizon-technical-challenges-regulatory
[77]European Medicines Agency (2020) EMA Regulatory Science to 2025: Strategic reflection. Amsterdam: European Medicines Agency. https://www.ema.europa.eu/en/documents/regulatory-procedural-guideline/ema-regulatory-science-2025-strategic-reflection_en.pdf
[78]European Union (2016) Consolidated Version of the Treaty on the Functioning of the European Union, art. 173 (ex art. 157 TEC). Official Journal C 202, 7 June 2016, p. 126. https://eur-lex.europa.eu/eli/treaty/tfeu_2016/art_173/oj/eng
[79]European Commission (2025) Commission Work Programme 2026 – Europe’s Independence Moment: Annexes 1–5 to the Communication from the Commission to the European Parliament, the Council, the European Economic and Social Committee and the Committee of the Regions, COM(2025) 870 final. Strasbourg, 21 October 2025. https://eur-lex.europa.eu/resource.html?uri=cellar:7f0c63c8-ae8f-11f0-89c6-01aa75ed71a1.0001.02/DOC_2&format=PDF
