
Clarifying the grey zone: a proposal
This is the last part of Between wellness and medicine, an article series examining the growing market of consumer neurotechnology devices caught between medical and wellness regulation.
Introduction
As we explored in the first three parts of this series, emerging neurotechnologies are increasingly blurring the line between wellbeing and healthcare. These devices often use the same technologies as clinical tools, but when marketed as wellness products, they bypass the rigorous testing required of medical devices and go straight to market. With more and more companies taking this route, the need for robust governance is becoming urgent.
Drawing on our extensive market research, an expert roundtable convened in early 2025, and interviews and discussions with a wide range of stakeholders — including leaders of startups and established companies — we propose a novel EU governance framework for this fast-growing category of health-adjacent consumer products.
Crucially, while developed in the context of consumer neurotechnology, this framework could apply to the broader family of wellness technologies, from fitness trackers and wellbeing apps to eye-tracking devices and AI therapists and companions.
This proposal is a starting point—an invitation to discussion, debate, and refinement. We see it as a way to build shared clarity before regulatory gaps widen further, and welcome all perspectives as the idea continues to evolve, along with the landscape.
Current regulatory challenges for wellness devices in the EU
Currently, there is no specific regulation for wellness devices in Europe — rather they are governed by a patchwork of multiple frameworks[1] (see our recent Neurotech governance map). As explained in Parts two and three of this series, this regulatory grey zone risks consumers potentially being misled, leaves businesses without clarity, and regulators unaware. Innovation also could be stifled in some areas, while risks could slip through the cracks in others.
Furthermore, wellness devices often process the same kinds of physiological or psychological data as medical devices, yet under GDPR the treatment of wellness data is ambiguous, and could be considered health data, biometric data, or simply personal data, depending on the framing[2],[3], creating uncertainty for companies and leaving consumers with uneven protection.
Marketing is where the grey zone is most visible. Many wellness products avoid medical regulation by using carefully chosen terms—such as “helps manage stress,” “supports sleep,” or “alleviates low mood.” These phrases stop short of formal medical claims like “treats anxiety” or “cures depression,” and thus fall outside the scope of the MDR (see Part 2 for examples).
All stakeholders we consulted — including those in the industry — agreed that health-adjacent wellness technologies cannot remain in this regulatory limbo. Without clearer rules and stronger oversight, Europe might forfeit opportunities for better prevention and care, undermine consumer trust, and compromise user safety.
With the recent reopening of the MDR, we initially wondered whether it could be adapted to encompass the wellness sector. But simply bringing wellness devices into scope of the MDR is not the answer, for two main reasons:
- The MDR’s definition of a medical device depends on an “intended medical purpose”, which many of these products do not—and should not—claim. The compliance burden on companies and notified bodies would thus be disproportionate.
- Business models and objectives in the consumer and medical markets differ fundamentally (see Part 3).
We therefore explored alternative options, starting by looking for parallels with other similarly borderline fields. Indeed, the EU already regulates other health‑adjacent products outside of medical law.
- Cosmetics are governed by Regulation (EC) No 1223/2009[4], which requires a safety assessment, strict labelling and draws line on acceptable marketing claims — without treating them as medical substances.
- Food supplements are covered by Directive 2002/46/EC[5], which mandates permissible marketing practices, as well as specific labelling.
- Nutrition and health claims on foods are tightly controlled under Regulation (EC) No 1924/2006[6], including a system to authorise health claims based on scientific evidence, a public register for all permitted claims, and establishment of nutrient profiles to inform consumers of their contents.
- For digital health, groundwork already exists. ISO/TS 82304‑2[7] defines a quality and reliability label for health and wellness apps, and the EU‑funded Label2Enable project[8] operationalised it for European use—useful building blocks for a proportionate scheme for wellness devices.
There are also examples of flexible oversight for borderline technologies and professions from around the world.
- Australia’s Therapeutic Goods Act exempts certain low‑risk software while still ensuring a degree of oversight, notably, some digital mental‑health tools[9].
- South Africa regulates health‑adjacent professions, such as chiropractors, under the Allied Health Professions Act[10], providing tailored oversight without classing them as medical.
- China’s recently released 2030 BCI roadmap focuses on breakthroughs and global leadership rather than drawing a line[11] between wellness and therapy, which further underscores the policy choices facing Europe.
This signifies that there are both parallels and precedents that justify the proposition for a novel legal framework to regulate wellness devices as an independent class of technologies from medical devices.
Design principles for clarifying the grey area
Policy action must balance evidence with anticipation when it comes to weighing up opportunities and risks of a new technology. In today’s political climate — where Europe is under pressure to strengthen its competitiveness in tech and innovation — this is an increasingly tricky balance to strike. Evidence on the impact of certain wellness neurotechnologies, either positive or negative, is still limited, as documented in our first two briefs. That means policymakers are forced to work without clear guardrails provided by scientific evidence.
In this situation, an anticipatory governance approach can offer guidance: proportionate, forward-looking policies that are open to revision, and informed by strategic intelligence and stakeholder engagement.[12]
Building on the principles set out in our 2024 publication Towards inclusive EU governance of neurotechnologies, we propose four core design principles to guide creation of a new framework in this sector:
- Grounded in technological reality. Oversight must be grounded in actual technological capabilities, trends, and limitations (lessons can be learned from the challenges caused to the research community by classing all non-invasive brain stimulation devices as medical under Annex XVI of the MDR[13]). Furthermore, it should be grounded in transparent risk assessment, accounting not only for physical risks but also for threats to human rights, as well as cognitive safety and integrity — the latter of which are not yet part of standard health technology risk assessments.
- Coherent and navigable. Companies need a clear and secure regulatory compliance path to innovate and succeed. According to our stakeholder interactions, the challenge in Europe is neither a lack of regulation, nor overregulation as such, but rather fragmentation and ambiguity. A framework for wellness technologies should mostly serve to bring together relevant rules (i.e. GDPR, MDR, AI Act, UCPD, EHDS) into a coherent package that clarifies what applies, when, to whom, and how. Definitions, requirements, and labels should align with scientific consensus and international principles, such as the WHO definition of health.
- Adaptive and proportionate. The digital health ecosystem changes fast. This framework needs to be flexible and responsive. It should avoid adding disproportionate burdens, especially for startups and SMEs, and instead act as a platform for oversight and quick reaction as the market evolves. Specifically, as the tech is increasingly based on software that is continuously updated, it is important to ensure that such a framework is flexible, adaptable, and responsive to emerging capabilities and features, and tailored to software applications, which most of these devices involve. Mechanisms such as delegated acts, regulatory sandboxes, and adverse-event monitoring could provide mechanisms for continuous adjustment, allowing oversight to evolve alongside new technologies and business models.
- Participatory and inclusive. Effective oversight requires feedback loops and inclusive dialogue, particularly in the field of neurotechnology which touches on very personal and intimate aspects of human nature and sense of self. A new framework should embed ways for stakeholders—researchers, developers, consumers, and policymakers—to contribute and respond to emerging developments. It should also ensure scientists are involved, as detailed definitions and a robust evidence-base are crucial. This proposal is only a starting point; the design must be debated, refined, and validated widely.
Proposal: a new EU framework for Wellness Devices
Across our stakeholder engagements, we have encountered broad support for creating a separate legal category for products that are not medical devices but are clearly health-adjacent: tools that promote wellbeing, help prevent decline, or support people with conditions, without diagnosing or treating disease. This category would go beyond neurotechnology to cover fitness trackers, digital wellness apps, AI therapists, eye-tracking tools, and other low-risk stimulation devices.
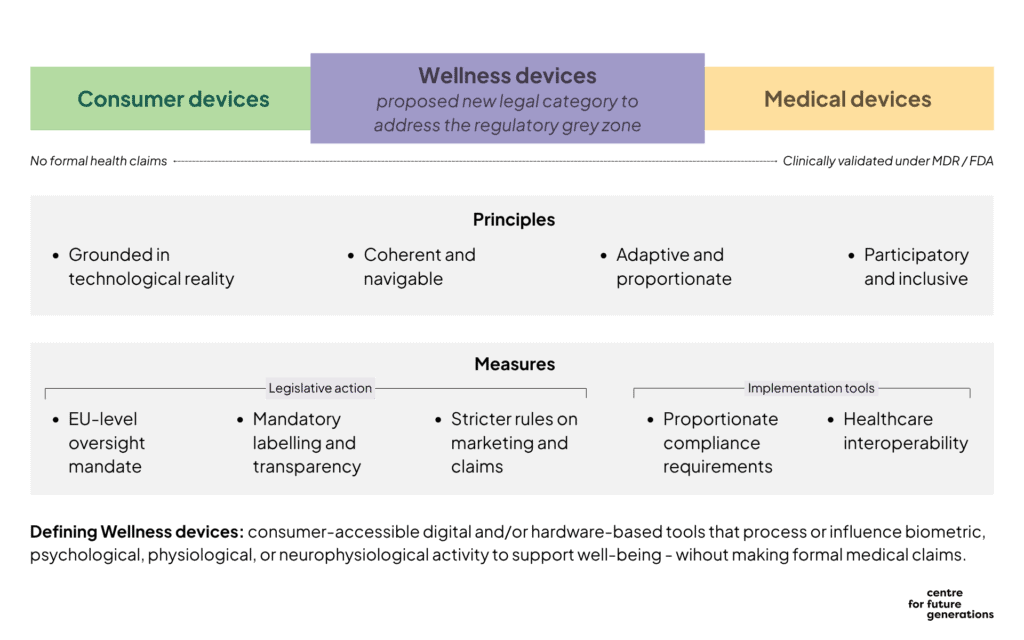
We propose a working definition:
“Wellness devices are consumer-accessible digital and/or hardware-based tools that process or influence biometric, psychological, physiological, or neurophysiological activity to support health, well-being, cognitive function, or symptom management, without making formal medical claims of diagnosis, prevention, or treatment.”
To govern this new category, we propose the following measures. The first three would likely require a legislative basis, whereas the final two could be advanced through implementing acts, working documents or codes of practice.
1. An EU-level oversight mandate
No public authority currently has a dedicated mandate to track this sector. Oversight must be harmonised at European level to avoid fragmentation across Member States, which would undermine both consumer protection and the Single Market. The Commission could establish a dedicated oversight body or expand the mandate of an existing agency to track the wellness devices market, issue certifications, and coordinate market surveillance. Alternatively, this could be an independent authority, led by a civil society organisation. Either way, this body would have responsibility for implementing the four points below, as well as:
- Issuing and informing guidance on marketing, transparency, and risk disclosure.
- Centralised adverse event reporting to improve accountability, particularly in a market with limited surveillance and enforcement capacity.
2. Mandatory labelling and transparency standards
Transparency builds consumer trust. Voluntary labelling schemes alone are unlikely to gain traction without legal backing or clear incentives, and there are risks of “label inflation”[14] if multiple standards emerge. Wellness devices should be subject to a standardised EU-wide labelling scheme covering efficacy, safety, data governance, and ethical design. This scheme could build on existing initiatives such as the EU-funded Label2Enable project (built on ISO 82304)[15] national equivalents like ORCHA[16]. Other initiatives include the upcoming NeuroTrust index in development by the World Economic Forum and Duke University[17] and the European Brain Council’s Seal of Responsible Neurotechnologies – a voluntary, independently verified ethical label operationalising the European Charter for the Responsible Development of Neurotechnologies.[18]
Unlike Medical Device certification, this is not a yes-or-no to enter the market, but rather it would provide consumers independent and standardised information on important considerations such as safety, efficacy, and data security of the device. To ensure cohesion, there should be a single scientifically-validated standard, with clear criteria for certification.[19]
3. Stricter rules on marketing and claims
Marketing is where the blurred line between wellness and healthcare is most problematic. Oversight should ensure that wellness device advertising is transparent, evidence-based, and cannot accidentally be perceived as medical to consumers, particularly in the mental health space where prospective users are especially vulnerable. Particular care should be taken towards transient platforms such as short-lived video feeds on social media, where health-adjacent claims are oftentimes more overt than on standing webpages and storefronts. The forthcoming Digital Fairness Act provides an opportunity to align wellness marketing standards with broader reforms of online consumer protection. Clearer rules on what can—and cannot—be claimed would protect consumers while preventing companies from overstating health benefits.
4. Proportionate and supported compliance requirements
A Wellness Devices legal framework should draw on relevant parts of existing regulation (e.g. GDPR, MDR, AI Act, the Unfair Commercial Practices Directive, the DSA, and the upcoming DFA) and translate them into practical, sector-specific guidance for the wellness sector. Standardised documentation and tools such as a code of practice and model consent forms could reduce compliance burden and guide development according to credible, science-based standards, provided they reduce rather than add to regulatory complexity—particularly for startups and SMEs. Regulatory sandboxes could allow companies to test products in controlled environments, refining compliance processes and supporting iterative product design. Crucially, enforcement also needs to be proportionate and effective — fines are currently the main enforcement tool, but they may not be effective as some companies may simply absorb them as a cost of doing business.
5. Interoperability with the healthcare sector
Wellness startups often want their technologies to meaningfully contribute to health outcomes — whether to enhance prevention of mental and neurological disorders, or by helping with symptom management, or even by sharing data with healthcare professionals to inform clinical intervention. Yet today, there are few routes for doing so.This legal framework should facilitate data flows into healthcare systems—while safeguarding privacy and security. The European Health Data Space (EHDS)[20] provides scope for an entry point of wellness devices in Electronic Health Records (see Articles 47 – 49), and conversations on this are beginning, including within the European Commission[21], but real integration will require dedicated infrastructure and coordination. Building a clear path for interoperability of wellness devices with the healthcare sector would allow them to contribute more meaningfully to prevention, early intervention, and personalised care within a holistic digital health landscape.
What needs to happen now
The framework we propose would bring benefits across the board. For startups, it would simplify compliance and provide the certainty needed to innovate. For consumers, it would raise trust, transparency, and safety. For regulators, it offers a pragmatic and proportionate tool to oversee a fast-moving sector. For the EU, it strengthens competitiveness by setting out a clear, science-based alternative to more rigid or fragmented approaches elsewhere. Most importantly, it would help unlock the potential of wellness technologies to support personalised prevention and management of neurological and mental health challenges, which currently has no dedicated infrastructure.
Yet, implementation questions of course remain. Decisions on who issues labels—government authorities or accredited independent bodies—will be critical. In addition, any framework should connect to reimbursement systems, EU innovation grants, and support schemes, ensuring uptake and fairness across the Single Market. Furthermore, as the design of enforcement architectures in the digital rule book illustrates, regulatory effectiveness ultimately depends not on the ambition of rules but on the credibility of their implementation and oversight mechanisms. Past failures in certification regimes[22] show the risks of weak oversight, capture, or box-ticking.
The first step to realising this framework would be the creation of an inter-DG task force or expert group to begin cross-policy coordination. As a recent Joint Research Centre study observed[23], there is currently no “home” for neurotechnology within the Commission—highlighting the need for a dedicated locus of expertise and coordination. The same can be said for wellness devices more generally, which span the competences of health, trade, consumer protection, and the single market.
The next step is to shape this framework collaboratively, with industry, researchers, policymakers, civil society, and citizens alike. The grey zone between wellness and medicine is too important to leave uncharted. Clearer rules today mean safer choices for consumers and stronger innovation for Europe tomorrow.
[1] OMC Medical. (2025). Smart Wearables Regulations in the EU. https://omcmedical.com/smart-wearables-regulations-in-the-eu
[2] Taka, AM. (2023). A deep dive into dynamic data flows, wearable devices, and the concept of health data. International Data Privacy Law, 13(2). https://academic.oup.com/idpl/article/13/2/124/7186915
[3] Istace, T. (2024). Neurodata: Navigating GDPR and AI Act Compliance in the Context of Neurotechnology. Geneva Academy. https://geneva-academy.ch/wp-content/uploads/2025/09/Neurodata-Navigating-GDPR-and-AI-Act-Compliance-in-the-Context-of-Neurotechnology.pdf
[4] European Union. (2009). Regulation (EC) No 1223/2009 of the European Parliament and of the Council of 30 November 2009 on cosmetic products (recast). https://eur-lex.europa.eu/eli/reg/2009/1223/oj/eng
[5] European Union. (2002). Directive 2002/46/EC of the European Parliament and of the Council of 10 June 2002 on the approximation of the laws of the Member States relating to food supplements. https://eur-lex.europa.eu/eli/dir/2002/46/oj/eng
[6] European Union. (2006). Regulation (EC) No 1924/2006 of the European Parliament and of the Council of 20 December 2006 on nutrition and health claims made on foods. https://eur-lex.europa.eu/eli/reg/2006/1924/oj/eng
[7] International Standards Association. (2021). Health software. Part 2: Health and wellness apps — Quality and reliability. https://www.iso.org/standard/78182.html
[8] Label2Enable. https://label2enable.eu/
[9] Australian Government. (2026). Software-based medical device exclusions. https://www.tga.gov.au/resources/guidance/understanding-if-your-software-based-medical-device-excluded-our-regulation
[10] South African Government. (1982). Allied Health Professions Act (previously Chiropractors, Homeopaths and Allied Health Service Professions Act and Associated Health Service Professions Act) 63 of 1982. https://www.gov.za/documents/associated-health-service-professions-act-20-mar-2015-1128
[11] Borklemans, D. (2025). China Unveils 2030 Brain-Computer Interface Roadmap. Neurofounders. https://www.neurofounders.co/articles/china-unveils-2030-brain-computer-interface-roadmap
[12] The OECD has created a Framework for Anticipatory Governance of Emerging Technologies, which calls for embedding societal values in innovation, using foresight and technology assessment, engaging stakeholders, building agile and adaptive regulation, and reinforcing international cooperation in science and standard-setting.
[13] Bublitz, C, Ligthart, S. (2024). The new regulation of non-medical neurotechnologies in the European Union: overview and reflection. Journal of Law and the Biosciences 11(2). https://academic.oup.com/jlb/article/11/2/lsae021/7774876
[14] Quote from a participant in our roundtable discussion.
[15] Label2Enable. https://label2enable.eu/
[16] ORCHA. https://orchahealth.com/
[17] World Economic Forum. (n.d.) Global Future Council on Neurotechnology. https://initiatives.weforum.org/global-future-council-on-neurotechnology/home
[18] European Brain Council (2026). Press release: EBC Introduces the Seal of Responsible Neurotechnologies. https://www.braincouncil.eu/ebc-seal-of-responsible-neurotechnologies/
[19] According to Apple Insider, Apple recently introduced a labelling system for certified medical device applications in its app store — another useful building block.
[20] European Union (2025). Regulation (EU) 2025/327 of the European Parliament and of the Council of 11 February 2025 on the European Health Data Space and amending Directive 2011/24/EU and Regulation (EU) 2024/2847. https://eur-lex.europa.eu/eli/reg/2025/327/oj/eng
[21] Joint Research Centre (2025). Wearable data: can they be shared for a public interest, such as healthcare? https://joint-research-centre.ec.europa.eu/jrc-news-and-updates/wearable-data-can-they-be-shared-public-interest-such-healthcare-2025-07-11_en
[22] For example: European Commission (2014). Joint plan for immediate actions under the existing medical devices legislation (PIP Action Plan). https://health.ec.europa.eu/system/files/2023-06/md_directive_pip-action-plan_commisson-swd.pdf
[23] Mochan et al. (2025). Emerging applications of neurotechnology and their implications for EU governance. https://publications.jrc.ec.europa.eu/repository/handle/JRC141928

